Product Images Lisdexamfetamine Dimesylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Lisdexamfetamine Dimesylate NDC 43547-606 by Solco Healthcare Us,llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 3 - image 04
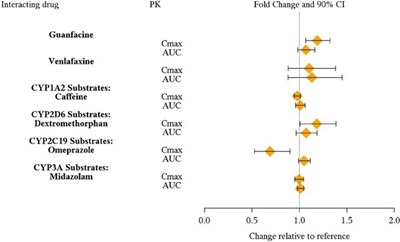
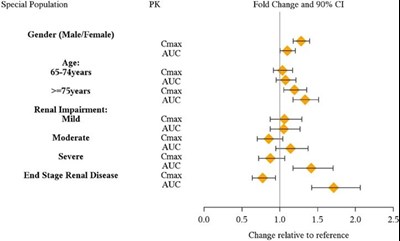
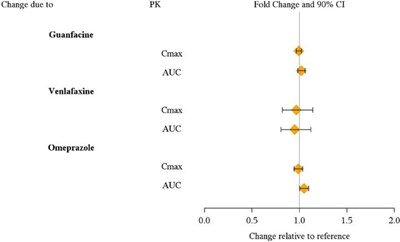
Interacting drug: Guanfacine, Venlafaxine CYP1A2 Substrates: Caffeine CYP2D6 Substrates: Dextromethorphan CYP2C19 Substrates: Omeprazole CYP3A Substrates: Midazolam PK parameters: Cmax and AUC Fold Change and 90% confidence interval (CI): - The fold change for the interaction is 0.0 - T T T indicates no significant change relative to the reference - The change relative to reference is 20%*
figure 4 - image 05
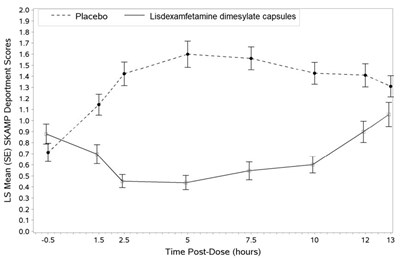
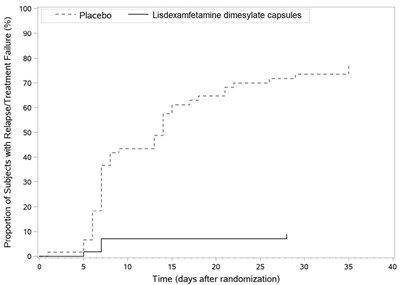
The given text appears to be a table showing LS Mean (SE) SKAMP Deportment Scores for a study involving a placebo and lisdexamfetamine dimesylate capsules. The table includes scores at different time points post-dose. However, the specific information regarding the scores and their interpretation is not available.*
20 - image 11

Each capsule contains 20 mg of Lisdexamfetamine dimesylate, which is equivalent to 11.6 mg of lisdexamfetamine. The recommended storage temperature is between 20°C and 25°C (68°F to 77°F), with permitted excursions between 15°C and 30°C (59°F to 86°F). The medication is defined in the USP. It is manufactured by Prinston Laboratories in Charlotte, NC and distributed by Solco Healthcare US LLC in Somerset, NJ. The package insert should be referred to for dosage information.*
40 - image 12

Each capsule contains 30 mg of Lisdexamfetamine dimesylate, which is equivalent to 17.3 mg of lisdexamfetamine. This medication should be stored at room temperature, between 20°C and 25°C (68°F to 77°F), with excursions permitted between 15°C and 30°C (59°F to 86°F). It is important to consult the package insert for specific dosage information. The medication is defined in the USP and is distributed by Solco Healthcare US, LLC, based in Somerset, NJ, USA. The manufacturer of the medication is Prinston Laboratories located in Charlotte, NC, USA. Unfortunately, no additional information is available from the provided text.*
50 - image 14

This text is a description of a medication called Lisdexamfetamine dimesylate. Each capsule contains 50 mg of the medication, which is equivalent to 28.9 mg of lisdexamfetamine. The recommended storage temperature is between 20°C and 25°C (68°F to 77°F). The medication is defined in the USP. Additional information, such as dosage instructions and distribution details, can be found in the package insert.*
70 - image 16

This text is a package insert for a medication called Lisdexamfetamine dimesylate. Each capsule contains 70 mg of the active ingredient, which is equivalent to 40.5 mg of lisdexamfetamine. The recommended dosage information is provided in the package insert. The medication should be stored at room temperature, between 20°C and 25°C (68°F to 77°F), with permitted excursions between 15°C and 30°C (59°F and 86°F). The medication is manufactured by Prinston Laboratories in Charlotte, USA, and distributed by Solco Healthcare USLLC in Somerset, USA. The insert mentions a separate medication guide provided to each patient. Unfortunately, the remaining part of the text is not legible.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.