Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- figure1 - Figure1
- figure2 - Figure2
- figure3 - Figure3
- figure4 - Figure4
- figure5 - Figure5
- figure6 - Figure6
- figure7 - Figure7
- carton1 - carton1
- carton2 - carton2
- carton3 - carton3
- carton4 - carton4
- carton5 - carton5
- carton6 - carton6
- carton7 - carton7
- carton8 - carton8
- container1 - container1
- container2 - container2
- container3 - container3
- container4 - container4
- container5 - container5
- container6 - container6
- container7 - container7
- container8 - container8
- figure10 - figure10
- figure11 - figure11
- figure12 - figure12
- figure9 - figure9
- structure1 - structure1
- structure2 - structure2
Product Label Images
The following 29 images provide visual information about the product associated with Pregabalin NDC 43598-292 by Dr.reddys Laboratories Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
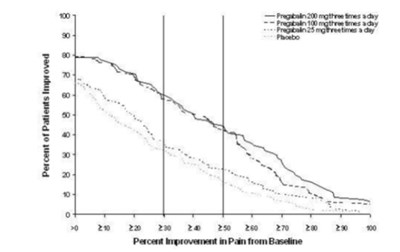
figure5 - Figure5

This text describes data related to the percent improvement in pain experienced by patients who were treated with Pregatalin 100 mg, Pregabalin 8 mg, and placeta. However, the information is incomplete and contains errors, making it difficult to provide a more detailed description. *
carton1 - carton1

The text describes a Unit Dose package of Pregabalin Capsules with 25mg strength. The package is not child-resistant and caution is advised to keep it away from children. The package contains 50 capsules in 5 x 10 unit-dose. The drug is intended for institutional use only and the NDC number is provided for the same. The rest of the text contains illegible characters and hence cannot be deciphered.*
carton2 - carton2

Pregabalin Capsules in 50 mg strength are packaged in a Unit Dose package that is not child-resistant and intended for institutional use only. The package contains 5 strips of 10 capsules each. The drug should be kept out of reach of children. The National Drug Code (NDC) is 43598-292-66. There is additional unreadable text.*
carton3 - carton3

This is a package of Pregabalin Capsules, 75mg strength, intended for institutional use only. The package contains 50 capsules (in 5x10 unit-dose form) manufactured by Dr. Reddys. The package is not child-resistant; hence it warns to keep it out of the reach of children. There is a unique product code NDC 43598-293-66 included for identification purposes. Apart from this, there seem to be some illegible characters and misplaced characters in the text, leading to random words and phrases.*
carton4 - carton4

This is a description of a medication called Pregabalin Capsules with a strength of 100mg. It is provided in a unit-dose package containing 50 capsules with each package showing the NDC code as 43598-294-66. The package is not child-resistant. The medication is only available with a prescription and should be kept out of the reach of children in a temperate storage condition. The origin of the medication is Dr. Reddy's Laboratories and the medication contains no more than 6 capsules of 100mg strength.*
carton5 - carton5

Pregabalin Capsules (150 mg) are intended for institutional use only and are packaged in unit-dose blister packs of 5 x 10 capsules (50 capsules total). The package is not child-resistant, so it is important to keep it and all drugs out of reach from children. The recommended dosage is one capsule per dose, and they should be stored at a controlled temperature between 20-25°C (68-77°F). The manufacturer is Dr. Reddy's and the product is Rx only. The rest of the text is not discernable and appears to be errors.*
carton6 - carton6

This is a package insert for Pregabalin Capsules, containing 200mg medication per capsule. It is intended for institutional use only and is not child-resistant. The pack comes with 50 capsules (5 x 10-unit dose) and should be kept out of reach of children. The medication must be stored at controlled room temperature. The NDC number is 43598-296-66. There is no mention of any side effects or prescriptions.*
carton7 - carton7

This is a prescription drug package of 50 capsules containing 225mg of Pregabalin USP, for institutional use only which is not child-resistant. It is advised to keep the medication out of reach of children. The NDC number of the package is 43598-297-66 and should be stored at controlled room temperature.*
carton8 - carton8

This is a description of Pregabalin Capsules provided in a unit dose package of 50 capsules (5 x 10 unit-dose) for institutional use only. Each capsule contains 300 mg of Pregabalin US. The package is not child-resistant so it should be kept out of reach of children. The recommended storage is at controlled room temperature.*
figure10 - figure10

This appears to be a table or graph with percentages of subjects without LTR (likely related to a medical study) for two groups - one that received Pregabain and another that received a placebo. The values start at 100% for the Pregabain group and decrease gradually while the values for the placebo group start at 0% and increase gradually. The chart spans over a period of 180 days.*
figure12 - figure12

This appears to be a graph or chart displaying the percent of subjects who improved from baseline to week 16. The data points range from 50 to 100 percent. However, since the text is not entirely clear and there is a lack of context, it is difficult to provide a more specific description.*
figure9 - figure9

This appears to be a chart or graph that displays the percentage of patients who experienced improvements in pain levels based on their daily dosage of Pregabalin and Progabalin, as well as a placebo. The x-axis shows different levels of improvement percentages while the y-axis shows the doses of Pregabalin and Progabalin. However, without additional context or a clear image, it is difficult to provide a more detailed description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.