Product Images Lurasidone Hydrochloride
View Photos of Packaging, Labels & Appearance
- Image - 290c7038 fd8c 4250 8569 99793e8864ea 01
- Image - 290c7038 fd8c 4250 8569 99793e8864ea 02
- Image - 290c7038 fd8c 4250 8569 99793e8864ea 03
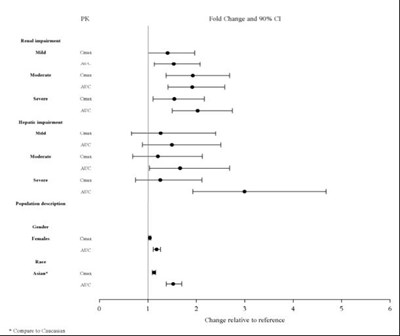
- Figure 3: Impact of Other Patient Factors on Lurasidone Hydrochloride Pharmacokinetics - 290c7038 fd8c 4250 8569 99793e8864ea 04
- container120mg - container120mg
Product Label Images
The following 5 images provide visual information about the product associated with Lurasidone Hydrochloride NDC 43598-355 by Dr. Reddys Laboratories Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - 290c7038 fd8c 4250 8569 99793e8864ea 02
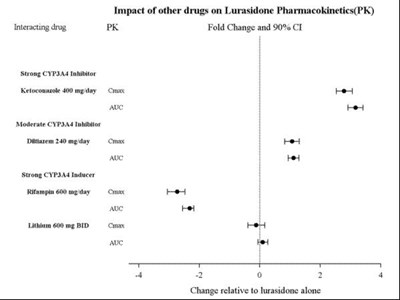
Impact of other drugs on Lurasidone Pharmacokinetics(PK). Interacting drug: Ketoconazole 400mg/day CYP3A4 inhibitor. Moderate CYP2D6 inhibitor: Not available. (The text appears incomplete and unclear to provide a useful description)*
Figure 3: Impact of Other Patient Factors on Lurasidone Hydrochloride Pharmacokinetics - 290c7038 fd8c 4250 8569 99793e8864ea 04
container120mg - container120mg

This is a package label containing information about its batch number, expiry date, serialization, and GTIN to be printed. The label shows an unvarnished area for imprinting the required information. It also has a long string of codes, symbols, and text arranged in rows and columns, which are not understandable and might be machine-readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

