Product Images Nitroglycerin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Nitroglycerin NDC 43598-436 by Dr. Reddys Laboratories Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
carton1 - carton1
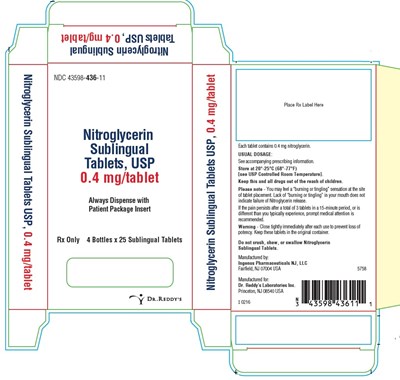
This is a product description for Nitroglycerin Sublingual Tablets USP, 0.4 mg/tablet. The tablets are dispensed in bottles of 25 and are intended for sublingual use only. Each tablet contains 0.4 mg of nitroglycerin. It is important to note that a "burning or tingling" sensation may occur upon tablet placement, and if pain persists after taking up to 3 tablets in a 15-minute period, prompt medical attention is recommended. The tablets should be stored at room temperature and kept out of reach of children. Additionally, the tablets should not be crushed, chewed, or swallowed. The manufacturer is Ingenus Pharmaceuticals NJ, LLC.*
carton2 - carton2
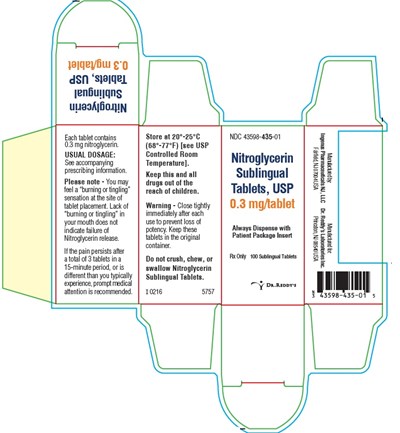
Each tablet of this product contains 0.3 mg of nitroglycerin and must be stored in a container at a controlled room temperature between 20°C to 25°C (68°F to 77°F). It is essential to read the prescribing information as these tablets can cause side effects such as tingling sensations. If the pain persists after taking a dosage, which can include 3 tablets after 15 minutes, it is recommended to seek medical attention. Nitroglycerin sublingual tablets should not be crushed, chewed or swallowed.*
carton3 - carton3
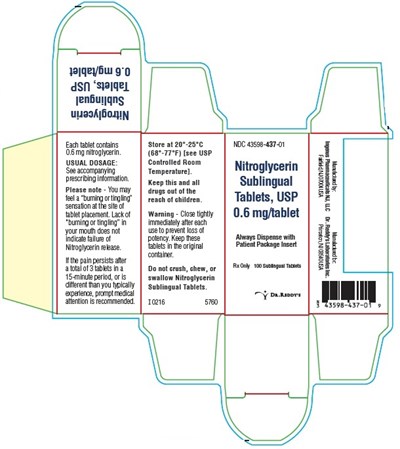
This is a medication label for Nitroglycerin Sublingual Tablets, USP 0.6 mg/tablet. It includes information on dosage, storage, and safety precautions. It also warns against chewing or swallowing the tablets and advises seeking medical attention if symptoms persist or worsen.*
container1 - container1

This is a Nitroglycerin Sublingual tablet with a strength of 0.4 mg per tablet. The NDC number for this product is 43508-436-35. It is recommended to always dispense with the patient package insert. However, the last line of the text is not readable and may contain non-English characters.*
container4 - container4

This is a description for Nitroglycerin Sublingual Tablets, 0.4 mg per tablet. It is available in a bottle of 100 sublingual tablets, with a prescription from Dr. Revov’s. The NDC number for this product is 43533-435-01. The other text seems to be unrelated and not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



