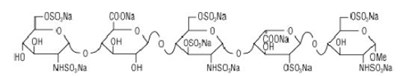
Product Images Fondaparinux Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Fondaparinux Sodium NDC 43598-608 by Dr. Reddy's Laboratories Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mg2sCarton - 10mg2sCarton

This appears to be a mix of random text and information on a medication called "Fondaparinux Sodium Injection" manufactured by "Gland Pharma Limited" and "Dr. Reddy's Laboratories Limited" in India. It comes in two pre-filled syringes and is used for subcutaneous injections. The text also includes some NDC codes and regulatory information. However, the majority of the text is not legible or does not seem to form any coherent meaning.*
2.5mg2scarton - 2.5mg2scarton

This is a product description for Fondaparinux Sodium Injection. It is a medication available in prefilled syringes for subcutaneous use and contains 25mg/0.5mL of the drug. The syringes are single-dose and have an active needle protection system. The manufacturer is Gland Pharma Limited and it is manufactured for Dr. Reddy's Laboratories Limited. The product is of Indian origin and the packaging dimensions are 152x28x88mm.*
5mg2scarton - 5mg2scarton

Fondaparinux Sodium Injection is a medication available in the form of prefilled syringes with an Active Needle Protection System. It is meant for subcutaneous use and must be dispensed by a pharmacist. The manufacturer is Gland Pharma Limited and is distributed by Dr. Reddy's Laboratories Limited. The package contains two 0.4mL prefilled syringes each with 5mg of injection. Unfortunately, the remainder of the text is not-available.*
7.5mg2scarton - 7.5mg2scarton

This is a description of a pharmaceutical product called "Fondaparinux Sodium Injection" that comes in two pre-filled syringes of 7.5mg/0.6mL or 75mg/0.6mL for subcutaneous use. The product is manufactured by Gland Pharma Limited for Dr. Reddy's Laboratories Limited. The package includes an NDC number of 43598-609.02 and a registered trademark of "Novaplus." The text also includes some illegible characters and numbers that seem to be part of a production code.*
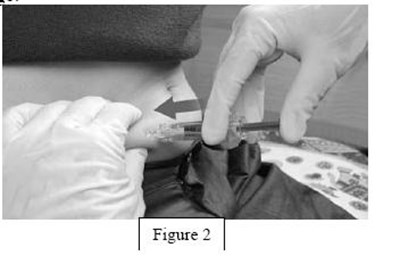
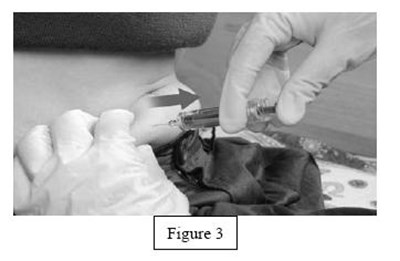
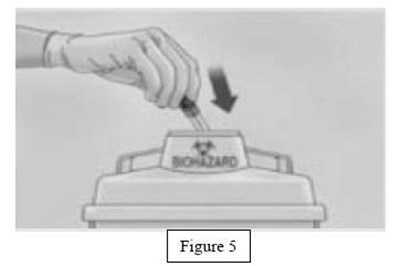
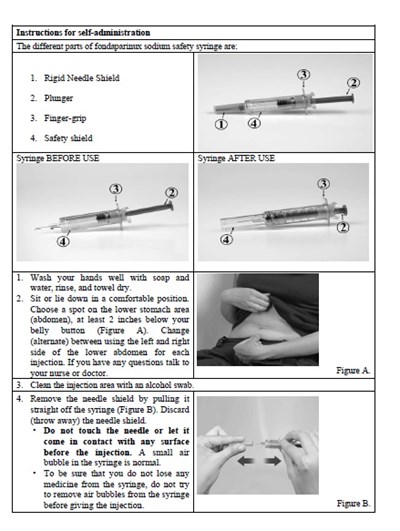
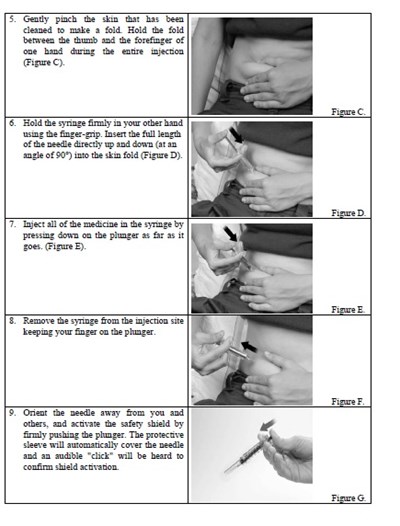
instrucn2cond - instruc2cond

This text provides instructions for the safe disposal of needles and syringes used for Fondaparinux Sodium injections. It advises to use a FDA-cleared sharps disposal container if available and to dispose of it properly when it is full. The container should be made of puncture-resistant plastic and labeled as hazardous. If a sharps disposal container is not available, a heavy-duty plastic container with a tight lid may be used. The community guidelines for safe sharps disposal should be followed. It is important not to recycle the used sharps disposal container and not to throw away needles and syringes in household trash.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.