Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Levetiracetam NDC 43598-637 by Dr.reddy's Laboratories Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1000carton - 1000carton

Levetiracetam is available in 0.75% Sodium Chloride Injection with a concentration of 1,000 mg/100 mL (1 m/mt). The rest of the text is not available or readable.*
1500 carton - 1500 carton
Levetiracetam is a medication that is injected into 0.54% sodium chloride solution at a concentration of 1500mg/100L (18 mrm). Unfortunately, without additional information, the purpose or specific use of this medication cannot be determined.*
500carton - 500carton
Levetiracetam is a medication that comes in the form of an injection mixed with 0.82% sodium chloride. Each injection contains 500 mg of the medication per 100 L. There is also a logo present on the packaging which reads "OrRoddys." The dimensions of the packaging are 265mm by 145mm.*
Figure1 - figure1
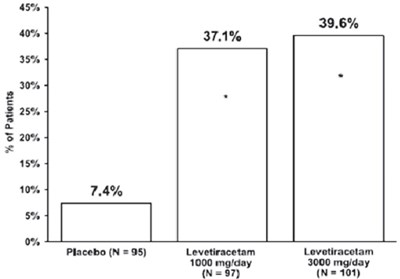
This is a chart showing the percentage of patients taking either a placebo or different doses of levetiracetam for a study. There were 95 patients taking the placebo, 97 taking 1000mg/day of levetiracetam, and 101 taking 3000mg/day of levetiracetam. The chart shows that 39.6% of patients taking the lower dose of levetiracetam experienced some benefit, while 37.1% of patients taking the higher dose experienced a benefit. 7.4% of patients taking the placebo experienced a benefit.*
Figure2 - figure2
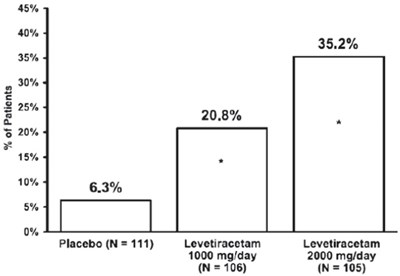
This appears to be a chart showing various percentages related to a study comparing a placebo to two different doses of levetiracetam (at 1000mg/day and 2000mg/day). The percentages listed may indicate the success rates or side effects associated with each treatment option. However, without more context or explanation, it is difficult to determine exactly what the percentages represent or how they were measured.*
Figure3 - figure3
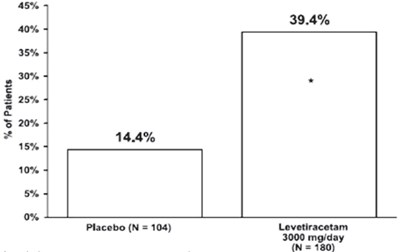
This appears to be a chart showing the percentage of patients who had a certain outcome or reaction to either a placebo (104 patients) or a medication called Levetiracetam (180 patients taking 3000 mg/day). The outcomes are not described and the chart includes some blank spaces.*
Figure4 - figure4
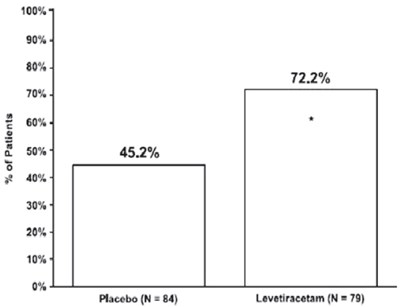
This text appears to be a table representing different percentages for two groups of data named "Placebo" (with 84 elements) and "Levetiracetam" (with 79 elements). The variable corresponding to these percentages is not mentioned, so it is not possible to determine what this table represents.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
