Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
- Figure-1.jpg - aripiprazole figure1
- Figure-2.jpg - aripiprazole figure2
- Figure-3.jpg - aripiprazole figure3
- Figure-4.jpg - aripiprazole figure4
- Figure-5.jpg - aripiprazole figure5
- Figure-6.jpg - aripiprazole figure6
- Figure 7 - aripiprazole figure7
- Figure 8 - aripiprazole figure8
- 10mg-30 - aripiprazole label1
- 15mg-30 - aripiprazole label2
- structural formula - aripiprazole structure
Product Label Images
The following 11 images provide visual information about the product associated with Aripiprazole NDC 43598-733 by Dr. Reddy's Laboratories Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure-1.jpg - aripiprazole figure1
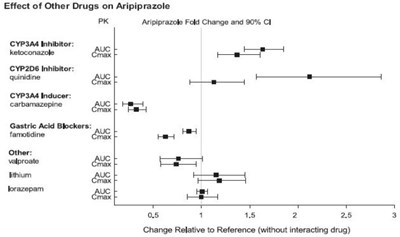
The text provides a table of the effect of different drugs on Aripiprazole, presented as fold change and 90% confidence interval. The drugs evaluated include CYP3A4 and CYP2DE inhibitors, CYP3A4 inducers, gastric acid blockers, and other drugs such as Viproste, lithium, and Lorazepam. The data indicates the change relative to a reference without the interacting drug.*
Figure-2.jpg - aripiprazole figure2

This is a table indicating the effect of different drugs on the medication Aripiprazole, with their corresponding PK values and their ability to inhibit or induce certain enzymes. The Dehydro-Aripiprazole Fold Change and 90% CI have also been provided, indicating the relative change in the medication's effects in the presence of these drugs.*
Figure-3.jpg - aripiprazole figure3
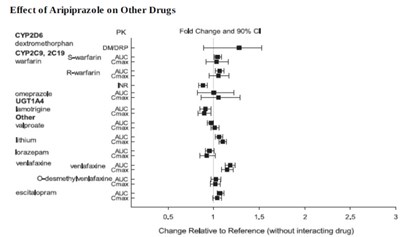
The text is discussing the effect of Aripiprazole, a drug, on the performance of other drugs such as dextromethorphan, Warfarin, Omeprazole, Venlafaxine, Lithium, and Lorazepam. The fold change and 90% C1 relative to the reference are given for each drug, but the actual values are not available.*
Figure-4.jpg - aripiprazole figure4
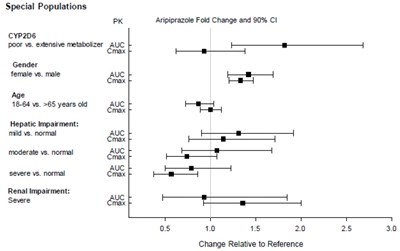
This is a table that seems to describe the effects of different conditions on the pharmacokinetics of a drug called aripiprazole. It looks at how factors such as genetics (CYP2D6 phenotype), gender, age, and liver or renal impairment affect the drug's maximum concentration (Cmax) and area under the curve (AUC). There also appears to be data on the change in aripiprazole levels and a confidence interval (90% CI). However, the purpose or context of this information is not available.*
Figure-5.jpg - aripiprazole figure5
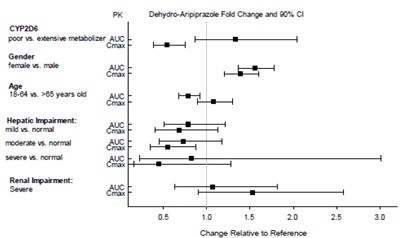
This appears to be a table displaying the fold change and 80% confidence interval of PK Dehydro-Aripiprazole for various factors such as CYP2D6 metabolizer status, gender, age, and hepatic/renal impairment. The values seem to compare the AUC and Cmax of different groups against a reference group. However, without further context or explanation, it is difficult to interpret the data meaningfully.*
Figure-6.jpg - aripiprazole figure6
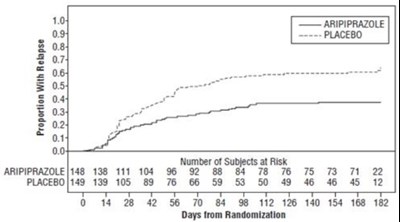
This appears to be a graph or table showing the proportion of subjects who experienced a relapse while taking Aripiprazole compared to a placebo. The table also includes the number of subjects at risk over time for both treatment groups. The x-axis appears to measure the number of days from randomization.*
Figure 8 - aripiprazole figure8
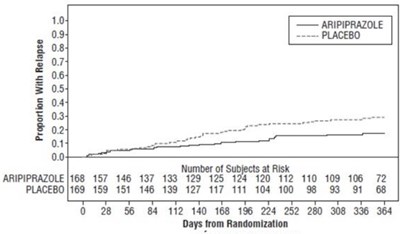
This appears to be a chart showing the proportion of subjects experiencing relapse over time for two groups: those receiving aripiprazole and those receiving a placebo. The chart also shows the number of subjects at risk at each time point. There is no additional information available.*
15mg-30 - aripiprazole label2

This text seems to be a mix of different types of data. It includes some distorted characters, special characters, symbols, some numerical data, and incomplete or unclear phrases. Therefore, it is not possible to generate a useful description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


