Product Images Famotidine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Famotidine NDC 43602-501 by Ascent Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
20 mg-30s - 20 mg 30s

This is a medication called Famotidine. It is a pack of 30 film-coated tablets, each containing 20 mg of Famotidine USP. The package insert contains information on the usual dosage. The medication is manufactured by Ascent Pharmaceuticals, Inc. in Central Islip, NY. It should be stored at a temperature between 20°C to 25°C and dispensed in a tight, light-resistant container. Excursions in temperature are permitted within the range of 15°C to 30°C. Rev: 12/20 is also mentioned.*
20 mg-500s - 20 mg 500s

This is a description of medication called Famotidine. It comes in the form of film-coated tablets and each tablet contains 20mg of Famotidine USP. It is prescribed by a doctor and the usual dosage can be found in the package insert. It should be stored at 20° to 25°C (68° to 77°F), but excursions are permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature]. The medication should be dispensed in a USP tight, light-resistant container. The manufacturer is Ascent Pharmaceuticals, Inc. located in Central Islip, NY 11722.*
40 mg-30s - 40 mg 30s

This is a description of Famotidine, a medication used to treat and prevent ulcers in the stomach and intestines. Each film-coated tablet contains 40 mg of Famotidine, and the usual dosage can be found in the package insert. It is to be stored at a temperature range of 20° to 25°C with excursions permitted to 15° to 30°C. It should be dispensed in a tight, light-resistant container. The medication is manufactured by Ascent Pharmaceuticals, Inc. in Central Islip, NY 11722.*
40 mg-500s - 40 mg 500s

This is information about Famotidine Tablets, USP, a prescription medication with 40mg of Famotidine USP per film-coated tablet. The package contains 500 tablets, and the usual dosage can be found in the package insert. The tablets should be stored between 20°C to 25°C (68°F to 77°F), with excursions permitted to 15°C to 30°C (59°F to 86°F). The medication should be dispensed in a USP tight, light-resistant container. Ascent Pharmaceuticals, Inc. in Central Islip, NY 11722 manufactures the tablets.*
Tab1 - Tab1
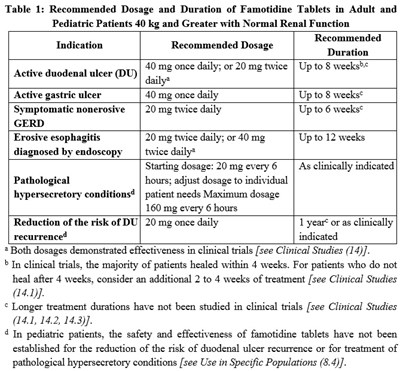
Table 1 provides the recommended dosage and duration of Famotidine Tablets for adult and pediatric patients. It includes the dosage and duration for various medical conditions such as GERD, active gastric ulcer, symptomatic nonerosive, erosive esophagitis diagnosed by endoscopy, and reduction of the risk of DU recurrence. Dosages range from 20mg once daily to a maximum of 160mg every 6 hours for pathological hypersecretory conditions. The efficacy of both dosages is discussed and the safety and effectiveness of famotidine tablets in pediatric patients is unknown.*
Tab2 - Tab2
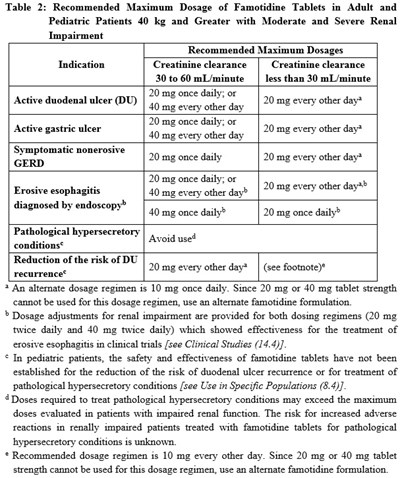
This is a dosage table showing the recommended maximum dosage of Famotidine tablets for adult and pediatric patients with moderate and severe renal impairment. The dosages vary depending on the indication and creatinine clearance levels. The table also includes alternate dosages for certain conditions, limitations on the use of some dosages for pediatric patients, and warnings about overdosing in patients with renal impairment.*
Tab3 - Tab3

The table presents serum concentrations of Famotidine associated with gastric acid reduction in Famotidine-treated pediatric and adult patients. The serum concentrations of Famotidine associated with 50% maximum gastric acid reduction are also presented as means = SD using the Sigmoid Eves model. The table includes data for healthy adult subjects and adult patients with upper GI bleeding.*
Tab4 - Tab4
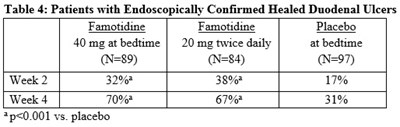
The table shows the percentage of patients with healed duodenal ulcers confirmed through endoscopy. The patients were treated with either Famotidine (40mg at bedtime or 20mg twice daily at bedtime) or Placebo for two and four weeks. At week 2, 2% of the Famotidine group and 3% of the 20mg twice daily Placebo group had healed ulcers, compared to 17% of the Placebo group. At week 4, none of the Famotidine group had ulcers, compared to 67% of the 20mg twice daily Placebo group and 31% of the placebo group. The statistical significance of the results is shown as 2p<0.001 (compared to placebo) which indicates that the findings are highly significant.*
Tab5 - Tab5
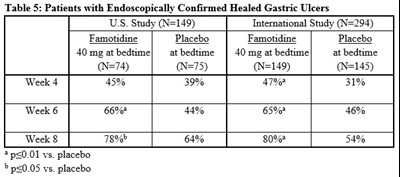
This text describes the results of two studies conducted on 149 and 294 patients with healed gastric ulcers respectively. The patients were given either 40mg of Famotidine or a placebo at bedtime for up to 8 weeks. The results showed that Famotidine was more effective in maintaining the healing of gastric ulcers compared to the placebo. These results were statistically significant, with Famotidine being significantly better than the placebo in preventing the recurrence of gastric ulcers.*
Tab6 - Tab6
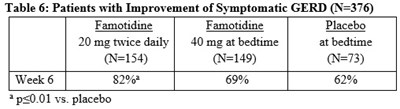
The text is a table showing the results of a study involving famotidine and placebo medication. Famotidine was given at a dose of 20 mg twice daily or 40 mg at bedtime, and the placebo was given to a group of patients. The study was conducted for 6 weeks, and the table shows that famotidine had a higher success rate compared to placebo. The results were significant with a p-value of 0.01.*
Tab7 - Tab7
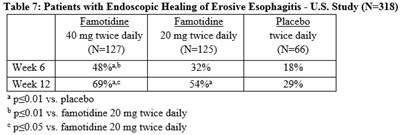
This is a table containing data on patients with erosive esophagitis who underwent endoscopic treatment with either Famotidine or a placebo. The table shows the percentage of patients who showed healing of the condition in week 6 and week 12 of the study. The results indicate that Famotidine was more effective than a placebo in healing the condition, and a higher dose of Famotidine was even more effective than a lower dose. The statistical significance of these results is also reported.*
Tab8 - Tab8

This is a table presenting data on patients with erosive esophagitis who received different dosages of Famotidine or Ranitidine. The table shows the percentage of patients who experienced endoscopic healing at week 6 and week 12 of treatment. Famotidine at 40 mg twice daily was found to be less effective than Famotidine at 20 mg twice daily and Ranitidine at 150 mg twice daily, with only 8% of patients experiencing healing after 6 weeks. The table also provides information on the statistical significance of the results.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
