Product Images Hizentra
View Photos of Packaging, Labels & Appearance
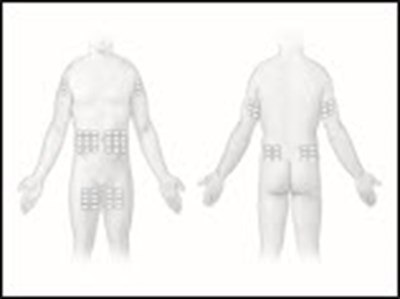
- Figure 1 - hizentra 01
- Figure 2 - hizentra 02
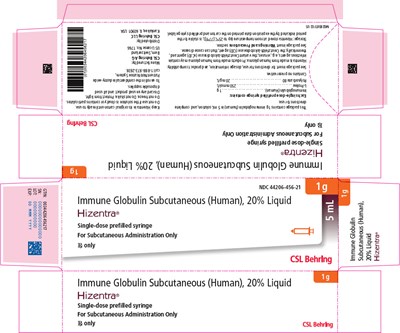
- Figure 3 - hizentra 03
- Figure 4 - hizentra 04
- Figure 5 - hizentra 05
- Figure 6 - hizentra 06
- Figure 7 - hizentra 07
- Figure 8 - hizentra 08
- Figure 9 - hizentra 09
- Figure 10 - hizentra 10
- Figure 11 - hizentra 11
- Figure 12 - hizentra 12
- Figure 13 - hizentra 13
- Figure 14 - hizentra 14
- Figure 15 - hizentra 15
- Figure 16 - hizentra 16
- Figure 17 - hizentra 17
- Figure 18 - hizentra 18
- Figure 19 - hizentra 19
- Figure 1 - hizentra 20
- Figure 1 - hizentra 21
- Figure 2 - hizentra 22
- Figure 3 - hizentra 23
- Figure 4 - hizentra 24
- Figure 5 - hizentra 25
- Figure 6 - hizentra 26
- Figure 7 - hizentra 27
- Figure 8 - hizentra 28
- Figure 9 - hizentra 29
- Figure 10 - hizentra 30
- Figure 11 - hizentra 31
- Figure 12 - hizentra 32
- Figure 13 - hizentra 33
- Figure 14 - hizentra 34
- Figure 15 - hizentra 35
- Figure 16 - hizentra 36
- Figure 17 - hizentra 37
- Figure 18 - hizentra 38
- Figure 19 - hizentra 39
- PRINCIPAL DISPLAY PANEL - 5 mL Vial Carton - hizentra 40
- PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton - hizentra 41
- PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton - hizentra 42
- PRINCIPAL DISPLAY PANEL - 50 mL Vial Carton - hizentra 43
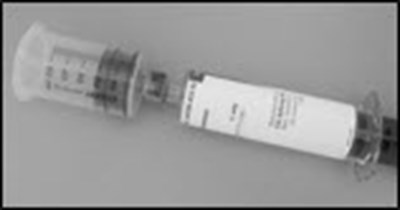
- PRINCIPAL DISPLAY PANEL - 5 mL Syringe Carton - hizentra 44
- PRINCIPAL DISPLAY PANEL - 10 mL Syringe Carton - hizentra 45
- PRINCIPAL DISPLAY PANEL - 20 mL Syringe Carton - hizentra 46
Product Label Images
The following 46 images provide visual information about the product associated with Hizentra NDC 44206-454 by Csl Behring Ag, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - hizentra 20

This appears to be a graph or chart with data points and labels on it. The graph shows some numbers and percentages related to a study, possibly on two different dosages of a medication (0.2 g/kg and 0.4 g/kg of Hizentra) versus a placebo. The numbers may refer to the probability of relapse or withdrawal for each treatment group over a period of time. Without more context or information, it is difficult to provide a full and accurate description of the data.*
PRINCIPAL DISPLAY PANEL - 5 mL Vial Carton - hizentra 40

This package contains a single-dose vial of Hizentra® immune globulin subcutaneous (human), which is a 20% liquid solution of human immunoglobulin. The complete directions for use, dosage information, and product compatibility are enclosed in the package insert. There is no preservative present in the solution. It is made from human plasma that may contain infectious agents and viruses, including the causative agent of Creutzfeldt-Jakob disease (CJD). The package insert lists all warnings and precautions associated with the use of Hizentra®. Storage of the product should be at room temperature up to 25°C (77°F) and kept in the original carton. There is a confidential industry program available and distributed by CSL Behring LLC.*
PRINCIPAL DISPLAY PANEL - 10 mL Vial Carton - hizentra 41

This is a product description of a 10 mL solution of Immune Globulin Subcutaneous (Human) with complete directions for use contained in the package which also includes single-dose vials. This human plasma-derived product contains no preservative and is 20% liquid. The product may cause CID (Creutzfeldt-Jakob disease) in rare instances and there is a warning and precautions section in the package insert for further information. The product should be stored at room temperature and the expiration date printed on the carton and label should be followed as well as protecting it from light. Any unused or expired product should be discarded along with used disposable supplies. There is a Patient Notification System available with a call to the manufacturer. The product is manufactured and distributed by CSLBehring.*
PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton - hizentra 42

This package appears to contain a medication called "Immune Globulin" in single-dose vials. The solution inside the vial is stable at room temperature for a period indicated by the expiration date. It is meant for subcutaneous administration. The package has directions for use and dosage information. There is also a phone number to enroll in a patient notification system.*
PRINCIPAL DISPLAY PANEL - 50 mL Vial Carton - hizentra 43

This is a description of CSL Behring's Hizentra - a single-dose subcutaneous Immunoglobulin (Human) containing 10g human immune globulin in 50L solution. The storage guidelines indicate that it should be stored at room temperature until the expiration date printed on the carton and the vial label. The package insert should be followed for usage directions, dosage information, and product compatibility. It is also noted that products made from human plasma may contain infectious agents such as viruses, theoretically causing diseases such as Creutzfeld-Jakob disease (CJD). Hence, a warning is given to refer to the package insert's Warnings and Precautions section.*
PRINCIPAL DISPLAY PANEL - 10 mL Syringe Carton - hizentra 45

The text contains some unreadable characters, no human understandable text is available.*
PRINCIPAL DISPLAY PANEL - 20 mL Syringe Carton - hizentra 46

This appears to be a medical description with the mentions of various drugs and their administration. The text also includes details about Immune Globulin Subcutaneous, 20% Liquid.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.