Product Images Valsartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
- Figure 1 - valsahctz figur1
- Figure 2-3-4 - valsahctz figure2 3 4
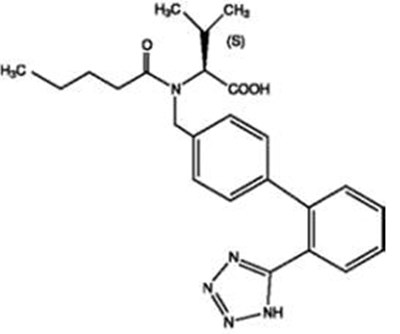
- Structure - valsahctz hctz
- Structure - valsahctz valsa
- 30's bottle pack - valsartan hctz 160125mg
- 30's bottle pack - valsartan hctz 16025mg
- 30's bottle pack - valsartan hctz 320125mg
- 30's bottle pack - valsartan hctz 32025mg
- 30's bottle pack - valsartan hctz 80125mg
Product Label Images
The following 9 images provide visual information about the product associated with Valsartan And Hydrochlorothiazide NDC 46708-456 by Alembic Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - valsahctz figur1

The text provided contains a chart showing the probability of achieving SBP (systolic blood pressure) lower than 140 mmHg at week 8, using different doses of certain medications. The medications listed are Valsartan and hydrochlorothiazide tablets with doses of 320/25mg and Hydralazine 25mg. The chart presents different baseline SBP values ranging from 140 to 180 mmHg.*
Figure 2-3-4 - valsahctz figure2 3 4

This document contains a chart showing the probability of achieving a certain blood pressure level after eight weeks of taking Valsartan and Hydrochlorothiazide tablets. There are graphs depicting the probability of achieving systolic and diastolic blood pressures below certain levels. Additionally, there is a table showing various blood pressure levels and the probability of achieving a diastolic blood pressure below 80 mmHg.*
30's bottle pack - valsartan hctz 160125mg

This is a description of a medication. The medication is in the form of a film-coated tablet containing 160mg of Valsartan USP and 125mg of Hydrochlorothiazide USP. The dosage should be determined according to the package insert. The medication should be stored at room temperature between 15-30°C (59-86°F), protected from moisture, in a tight container. The accompanying full Prescribing Information should be reviewed. The medication should be kept out of reach of children. The medication is manufactured by Alembic Pharmaceuticals Limited in Gujarat, India. The medication has a National Drug Code (NDC) of 46708-454-30 and comes in packages of 30 tablets. The medication is prescription-only and must be dispensed with an attached Patient Information Leaflet. The lot and expiration date are not available.*
30's bottle pack - valsartan hctz 16025mg

Ezch fim is a medication in tablet form that contains 160mg of Valsartan USP and 25mg of Hydrochlorothiazide USP. It is recommended to store the medication at a temperature of 25°C, and to protect it from moisture in a tightly sealed container. Manufactured by Aembic Pharmaceuticals Limited, this product is only available through prescription and comes with an accompanying patient information leaflet. The NDC number provided is 46708-455-30.*
30's bottle pack - valsartan hctz 320125mg

This text describes the details of a medication called Ezch that comes in the form of coated tablets. The tablet contains 320 mg of Valsartan USP and 125 mg of Hydrochlorothiazide USP. The package insert should be referred to for dosage instructions. The medication should be stored at a temperature of 25°C (T7°F), with permitted excursions to 15-30°C (59-86°F), and protected from moisture. It should be dispensed in a tight container, and the accompanying prescribing information should be referred to. The medication should be kept out of the reach of children. The manufacturing company Aembic Pharmaceuticals Limited, Formulation Division is located in Gujarat, India. The medication is available as 30 tablets, and the pharmacist should dispense it with attached patient information leaflet. Lot and Mig. Lc.No. is also mentioned.*
30's bottle pack - valsartan hctz 32025mg

This is a prescription drug called Valsartan and Hydrochlorothiazide Tablets, USP, which contains 320mg of Valsartan USP and 25mg of Hydrochlorothiazide USP. It should be stored at room temperature, with allowed excursions, and protected from moisture. The drug should be dispensed in a tight container and come with an accompanying prescribing information leaflet. It is manufactured by Alembic Pharmaceuticals Limited in India and has an NDC of 46708-457-30. It comes in a package of 30 tablets and is for prescription use only.*
30's bottle pack - valsartan hctz 80125mg

This is a prescription drug called Valsartan and Hydrochlorothiazide Tablets. Each coated tablet contains 80mg of Valsartan USP and 125mg of Hydrochlorothiazide USP. It is manufactured by Alembic Pharmaceuticals Limited and is dispensed in a tight container. The tablets should be stored at 25°C with a permitted excursion to 15-30°C and protected from moisture. The dosage should be referred to from the package insert. The full prescribing information is included in the package. The drug should be kept out of the reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.