Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- chemical-structure - chemical structure
- formula - formula
- spl pregabalin figure 8
- spl-pregabalin-figure1 - spl pregabalin figure1
- spl-pregabalin-figure10 - spl pregabalin figure10
- spl-pregabalin-figure11 - spl pregabalin figure11
- spl-pregabalin-figure12 - spl pregabalin figure12
- spl-pregabalin-figure3 - spl pregabalin figure3
- spl-pregabalin-figure4 - spl pregabalin figure4
- spl-pregabalin-figure9 - spl pregabalin figure9
- spl-pregabalin-label-100mg - spl pregabalin label 100mg
- spl-pregabalin-label-150mg - spl pregabalin label 150mg
- spl-pregabalin-label-200mg - spl pregabalin label 200mg
- spl-pregabalin-label-225mg - spl pregabalin label 225mg
- spl-pregabalin-label-25mg - spl pregabalin label 25mg
- spl-pregabalin-label-300mg - spl pregabalin label 300mg
- spl-pregabalin-label-50mg - spl pregabalin label 50mg
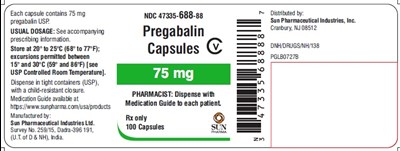
- spl-pregabalin-label-75mg - spl pregabalin label 75mg
Product Label Images
The following 18 images provide visual information about the product associated with Pregabalin NDC 47335-693 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
spl pregabalin figure 8

The text represents the responder rates in percentage for three different groups. The first group has a rate of 40.6%, the second group has a rate of 29.1%, and the third group has a rate of 22.6%. There is also a table with three columns representing "Placebo," "2.5 mg/kg/day," and "10 mg/kg/day," with row numbers and participant counts for each group.*
spl-pregabalin-figure1 - spl pregabalin figure1

The image shows a graph labeled as "Figure 1" from a study labeled as "DPN 1". The different lines on the graph represent the percentage of patients who achieved various levels of improvement in pain intensity. The x-axis represents the percent improvement in pain intensity from baseline, while the y-axis represents the percentage of patients improved. The four lines on the graph represent different treatment groups, including pregabalin 200mg, pregabalin 100mg, pregabalin 25mg, and placebo. This graph can be used to compare the effectiveness of these treatments in reducing pain intensity.*
spl-pregabalin-figure10 - spl pregabalin figure10

The figure shows the time taken for subjects to experience a loss of therapeutic response to pregabalin compared to placebo in a study on fibromyalgia (F2). The graph is a Kaplan-Meier analysis and displays the estimated percentage of subjects who did not experience a loss of therapeutic response over time. Not available.*
spl-pregabalin-figure12 - spl pregabalin figure12

The text describes a figure labeled "Figure 12" that shows the percentage of subjects who achieved different levels of improvement in pain intensity in a study called "SCI 2". The study was conducted over 16 weeks, and data is provided for both a drug called "pregabalin" and a placebo. The figure shows that improvement in pain intensity was greater in the pregabalin group compared to the placebo group.*
spl-pregabalin-figure3 - spl pregabalin figure3

The image shows the progress of patients on different doses of Pregabalin in Study PHN1 regarding the improvement in their pain intensity levels. The percentage of patients achieving various levels of pain improvement from baseline is depicted in the graph. There are three doses of Pregabalin: 300mg twice daily, 150mg twice daily, and 75mg twice daily. The term "Flacsbo" likely refers to the placebo group.*
spl-pregabalin-figure4 - spl pregabalin figure4

The text describes Figure 4 which shows the percentage of patients achieving different levels of improvement in pain intensity in a study called PHN 2. The study compared the effects of pregabalin at two different doses (200 mg and 100 mg taken three times a day), placebo was also used. The x-axis shows the percent improvement in pain from baseline, while the y-axis is not provided.*
spl-pregabalin-figure9 - spl pregabalin figure9

The figure shows the percentage of patients in a study (Fibromyalgia Study F1) who achieved various levels of improvement in pain intensity. The study compared three different daily doses of pregabalin (600mg, 450mg, and 300mg) with a placebo. The x-axis shows the percentage of patients improved, ranging from 0% to 100%. The y-axis shows the treatment groups: pregabalin 600mg, pregabalin 450mg, pregabalin 300mg, and placebo.*
spl-pregabalin-label-100mg - spl pregabalin label 100mg
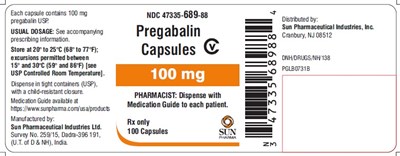
This is a medication distributed by Sun Pharmaceutical Industries, Inc. The usual dosage must be taken as indicated in the accompanying prescribing information. The medication must be stored within the temperature range of 20 to 25°C (66 to 77°F), and excursions between 15° and 30°C (59° and B6°F) are allowed. It should be dispensed in a tight container and accompanied with a medication guide for each patient. The manufacturing company is Sun Pharmaceutical Industries located in India.*
spl-pregabalin-label-200mg - spl pregabalin label 200mg

Each capsule contains 200 mg of pregabalin USP manufactured and distributed by Sun Pharmaceutical Industries, Inc. The usual dosage of Pregabalin is provided in the accompanying prescription. Store the capsules at temperatures between 20°C to 25°C. Dispense with medication guide to each patient in a tight container with a child-resistant closure. Pharmaceutical professionals can find the medication guide at Rx only @https://surpharma.com/usapodus.*
spl-pregabalin-label-225mg - spl pregabalin label 225mg

This is a description of a medication containing 225mg of pregabalin USP, distributed by Sun Pharmaceutical Industries, Inc. The usual dosage and prescribing information are not available. It should be stored between 20° to 25°C, with permitted excursions between 15° and 30°C, and dispensed in tight containers with a child-resistant closure. A medication guide is available at the listed website, and pharmacists are advised to dispense with the guide to each patient. The product comes in a package of 100 capsules.*
spl-pregabalin-label-25mg - spl pregabalin label 25mg

Each capsule of this medicine contains 25mg of Pregabalin USP. The usual dosage information is available in the accompanying prescribing information. The capsules should be stored between 15-30°C (59-86°F). Dispense it with a medication guide available at https://www.sunpharma.com/usa/products. The capsules should be tightly closed in child-resistant containers. The medicine is manufactured by Sun Pharmaceutical Industries Ltd., located at Survey No. 259/15, Dahanu Road - 401 601, Dist. Palghar (State of Maharashtra), India, and is distributed by Sun Pharmaceutical Industries, Inc. at Cranbury 08512.*
spl-pregabalin-label-300mg - spl pregabalin label 300mg

Each capsule of this medication contains 300g of pregabalin USP, and it is dispensed with a child-resistant closure. The usual dosage is indicated in the accompanying prescription note. The medication should be stored between 20° to 25°C (68° to 77°F) with excursions permitted between 15° and 30°C (59° and 86°F). It is important to refer to the USP Controlled Room Temperature. The medication guide for this medicine can be found on the manufacturer's website. The manufacturer is Sun Pharmaceutical Industries Ltd, located in India.*
spl-pregabalin-label-50mg - spl pregabalin label 50mg

This is a medication prescription information for Pregabalin capsules. The recommended dosage information is not listed and the text provides instructions for storage and dispensing. The medication should be stored between 20-25°C and excursions between 15-30°C are allowed. Dispensing should be done in tight containers with child-resistant closures and a medication guide should be provided to each patient. The name of the manufacturing company is mentioned as Sun Pharmaceutical Industries Ltd.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.