Product Images Zynrelef
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 86 images provide visual information about the product associated with Zynrelef NDC 47426-302 by Heron Therapeutics, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
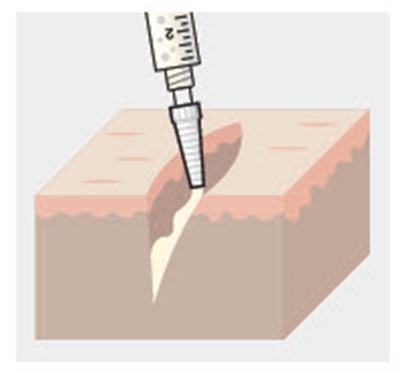
Figure 1 - zynrelef 04
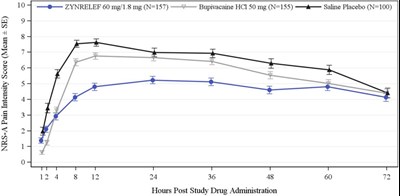
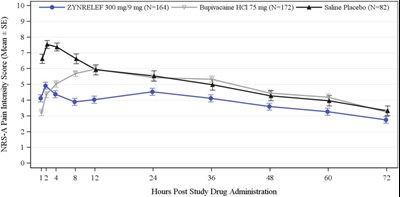
The text describes the NRS-A pain intensity score with mean and standard error for three different groups treated with ZYNRELEF, Bupivacaine HCI, and Saline Placebo respectively. The data is represented in a graph that shows the pain intensity score at different hours post-study drug administration.*
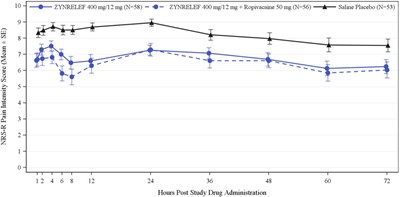
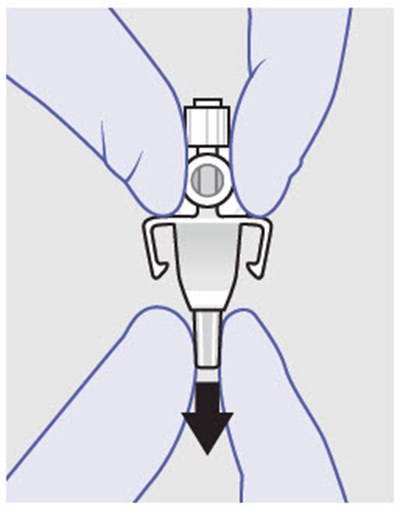
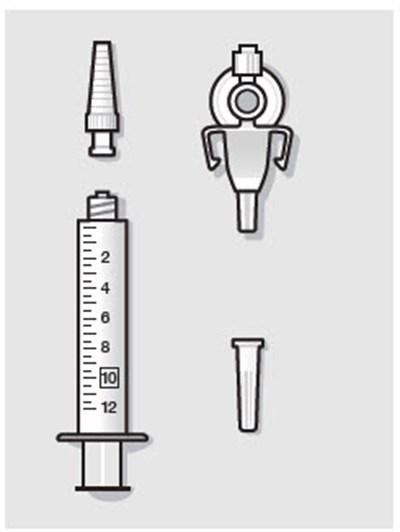
Figure - zynrelef 26

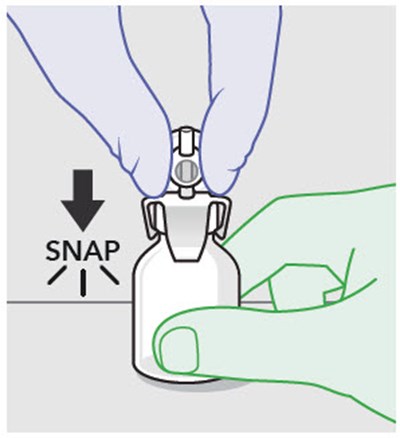
This text suggests that when assembling or replacing components, it is important not to use substitutes for any of the original parts. This is likely a safety precaution to ensure the proper functioning of the machine or device in question.*
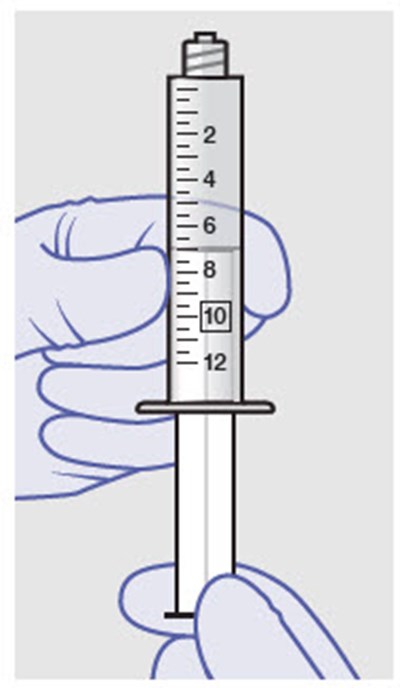
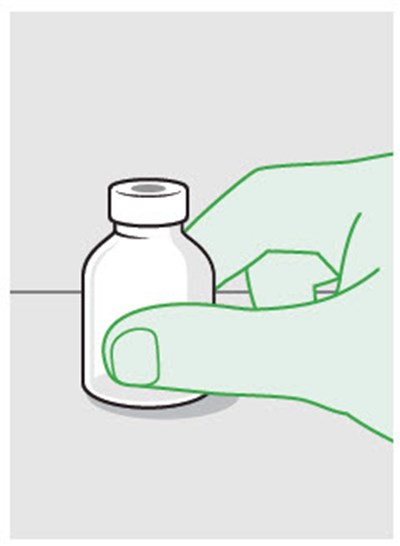
Figure - zynrelef 45

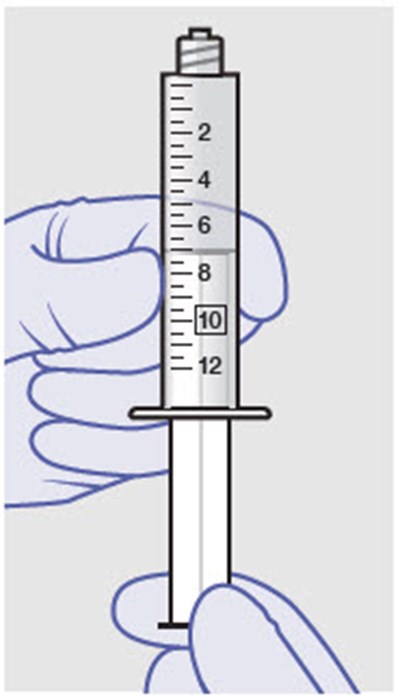
This is a warning message advising not to replace any of the parts or components.*
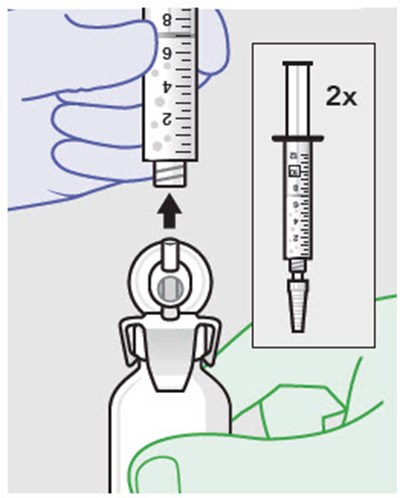
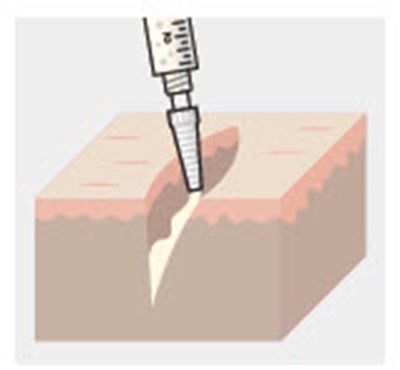
Figure - zynrelef 64
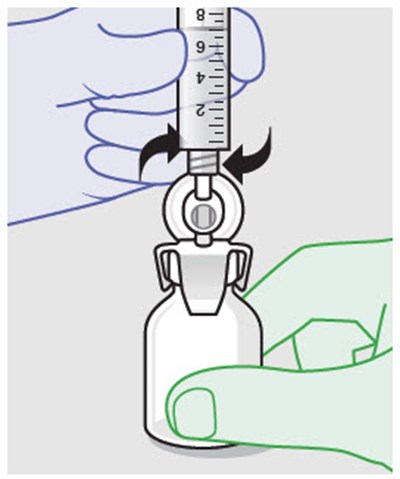
This text is readable and the description would be: "This text provides a warning not to substitute any of the components."*
PRINCIPAL DISPLAY PANEL - 2.3 mL Vial Carton Kit - zynrelef 86

ZYNRELEF is a solution for soft tissue or periarticular instillation use that contains 50 mg bupivacaine and 1.8 mg meloxicam. This single-dose application package includes a 23 mL vial and sterile syringe with an Luer Lock, cap, and applicator. The kit must be used as instructed, and unused portion must be discarded. Storage instructions mandate storing the kit from 2°C to 25°C. This text also includes batch and manufacturing information, along with prescribing and dosage instruction warnings.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.