Product Images Afrezza
View Photos of Packaging, Labels & Appearance
- Figure 3. Mean Insulin Effect and Pharmacokinetic profiles - Figure 3 PKPD
- IFU Know Your Cartridges
- IFU Open Packages
- IFU Room Temp Storage
- IFU Store Refrigerated
- Figure 1 Mealtime AFREZZA Starting Dose Conversion Table - afrezza 01
- Figure 2 - Mean Change in FEV - afrezza 02
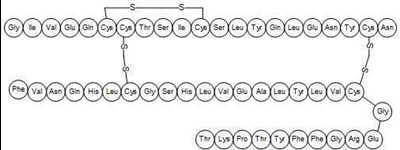
- Chemical Structure - afrezza 03
- Figure A - Image of Cartridges - afrezza 05
- Diagram of Inhaler - afrezza 06
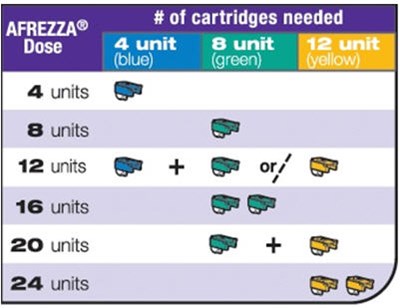
- Cartridge selection based on color - afrezza 08
- Figure B - Dosage Chart - afrezza 09
- Example combination - afrezza 10
- Image of pushing cartridge out of blister - afrezza 12
- Cartridges and inhaler should be at room temperature - afrezza 13
- Image of inhaler in hand - afrezza 14
- Image of inhaler being opened - afrezza 15
- Image of cartridge being placed into inhaler - afrezza 16
- Once loaded keep inhaler level - afrezza 17
- Image of inhaler being closed - afrezza 18
- Image of mouthpiece cover being removed - afrezza 19
- Check you are ready to inhale - afrezza 20
- Image of exhaling - afrezza 21
- Image of inhaler position in mouth - afrezza 22
- Image of deep inhale - afrezza 23
- Image of mouthpiece cover being replaced - afrezza 24
- Image of inhaler being opened - afrezza 25
- Image of cartridge being removed - afrezza 26
- Image of cartridge being thrown away - afrezza 27
- Image of 3 cartridges - afrezza 28
- Do not mix used and new cartridges - afrezza 29
- Before use cartridges should be at room temperature - afrezza 32
- Inhaler storage instructions - afrezza 33
- Replace inhaler every 15 days - afrezza 34
- Powder residue is normal; do not wash inhaler - afrezza 35
- Image of caution symbol - afrezza 36
- Dose conversion chart - afrezza 37
- Dose conversion chart - afrezza 38
- primary panels NDC 47918 874 90
- PRINCIPAL DISPLAY PANEL - 90 – 8 Unit Kit Carton - primary panels NDC 47918 878 90
- Primary Panels NDC 47918-880-18 - primary panels NDC 47918 880 18
- PRINCIPAL DISPLAY PANEL - 90 – 12 Unit Kit Carton - primary panels NDC 47918 891 90
- primary panels NDC 47918 898 18
- Primary Panels NDC 47918-902-18 - primary panels NDC 47918 902 18
Product Label Images
The following 44 images provide visual information about the product associated with Afrezza NDC 47918-898 by Mannkind Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
IFU Know Your Cartridges

The text describes the packaging and strengths of AFREZZA® cartridges. The package contains 1 foil package with 2 blister cards and 3 cartridges per package per strip. Each blister card has 5 strips. There are two strengths available: 4 units with one blue cartridge, and 8 units with one green cartridge.*
IFU Room Temp Storage

This is a set of instructions for storing and using opened foil packages of strips. The chart describes how long the strips can be used after opening depending on whether they are sealed or unsealed. It is recommended to not put a blister card or strip back into the refrigerator after being stored at room temperature.*
IFU Store Refrigerated

The text provides instructions for storing drugs in a refrigerated environment with a recommended temperature range of 36-46°F (2-8°C). The drugs should be kept in unopened foil packages and can be used until the expiration date or within one month of being opened. The text also mentions that the refrigerated storage is not in use.*
Figure 1 Mealtime AFREZZA Starting Dose Conversion Table - afrezza 01
This text seems to be a table showing the number of cartridges needed for different unit ranges. The table is divided into two columns: "4 unit" and "8 unit". Additionally, there is a row with the label "# of cartridges needed". The table also specifies different ranges of units from "up to 4 units" to "21-24 units" and the corresponding number of cartridges required for each range of units.*
Diagram of Inhaler - afrezza 06

The text appears to be a list of items related to some product. It includes a purple, removable cover for a mouthpiece, a white mouthpiece, a cartridge (presumably for the product), a white cartridge and a cup. The context of the product and its purpose is not clear from the text.*
Cartridges and inhaler should be at room temperature - afrezza 13

This text provides instructions for the use of cartridges and inhalers. Specifically, it advises the user to allow them to come to room temperature for 10 minutes prior to use.*
Once loaded keep inhaler level - afrezza 17

This text provides instructions on how to properly handle an inhaler after loading the cartridge to avoid losing drug powder. It emphasizes the importance of keeping the inhaler level and avoiding turning, pointing, shaking or dropping it. If any of these mishaps occur, the cartridge should be discarded and a new one loaded.*
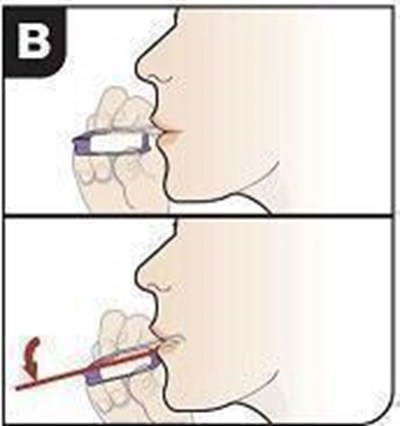
Check you are ready to inhale - afrezza 20

Check that you are ready to inhale: Purple mouthpiece cover is removed. Inhaler is held level. Fully review the following A-B-C steps prior to beginning the inhale process.*
Do not mix used and new cartridges - afrezza 29

This text provides instructions on how to properly handle cartridges. It urges caution when using new and used cartridges together and explains how to identify a used cartridge through the position of the white cup.*
Before use cartridges should be at room temperature - afrezza 32

This text provides a usage instruction for cartridges and inhaler stating that they should be at room temperature for 10 minutes before use.*
Inhaler storage instructions - afrezza 33

This text provides instructions for the storage of an inhaler. After taking a full dose, the purple mouthpiece cover should be replaced and the inhaler should be closed. The inhaler should be kept in a clean and dry place with the mouthpiece. It can be stored in the refrigerator, but must be at room temperature before use. The cartridges should never be left or stored in the inhaler, and the inhaler should be kept out of reach of children.*
Replace inhaler every 15 days - afrezza 34

This text refers to a time interval of fifteen days. No additional information is provided.*
Powder residue is normal; do not wash inhaler - afrezza 35

This text is a set of instructions for using and maintaining an inhaler. It explains that powder residue inside the mouthpiece is normal and doesn't affect the dose, and advises not to wash the inhaler but rather wipe the outside with a clean, dry cloth if necessary.*
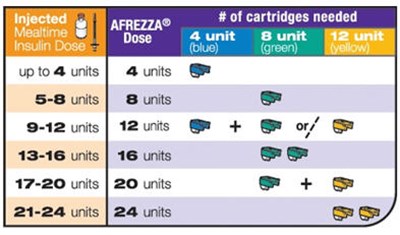
Dose conversion chart - afrezza 38

To switch from injected mealtime insulin to AFREZZA®, follow the chart to determine the appropriate dose based on your current insulin dosage. The chart provides a range of 5-24 units with corresponding mealtime insulin dosages.*
primary panels NDC 47918 874 90

The text contains some unrecognizable characters and random words, thus making it not-available for generating a useful description.*
PRINCIPAL DISPLAY PANEL - 90 – 12 Unit Kit Carton - primary panels NDC 47918 891 90

This appears to be a medication packaging label that provides information about the contents of the carton, including 90 cartridges and 2 inhalers. Dispensing instructions are included to provide a medication guide to each patient. However, some characters are not readable or are garbled, making it difficult to determine the medication's name or intended use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.