Product Images Duloxetine
View Photos of Packaging, Labels & Appearance
- d633baad 41fe 491c a563 517d726028c6 01
- d633baad 41fe 491c a563 517d726028c6 02
- d633baad 41fe 491c a563 517d726028c6 03
- d633baad 41fe 491c a563 517d726028c6 04
- d633baad 41fe 491c a563 517d726028c6 05
- d633baad 41fe 491c a563 517d726028c6 06
- d633baad 41fe 491c a563 517d726028c6 07
- d633baad 41fe 491c a563 517d726028c6 08
- d633baad 41fe 491c a563 517d726028c6 09
- d633baad 41fe 491c a563 517d726028c6 10
- d633baad 41fe 491c a563 517d726028c6 11
- d633baad 41fe 491c a563 517d726028c6 12
- d633baad 41fe 491c a563 517d726028c6 13
- d633baad 41fe 491c a563 517d726028c6 236 2
- d633baad 41fe 491c a563 517d726028c6 41 1
- d633baad 41fe 491c a563 517d726028c6 41 2
- d633baad 41fe 491c a563 517d726028c6 41 3
Product Label Images
The following 17 images provide visual information about the product associated with Duloxetine NDC 49252-007 by Inventia Healthcare Limited., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
d633baad 41fe 491c a563 517d726028c6 03

This appears to be a graph showing the proportion of patients with relapse over time (measured in days) following randomization. The y-axis shows the proportion of patients (ranging from 0% to 8%) and the x-axis shows the time in days. However, without more context or information it is difficult to provide a more detailed or accurate description.*
d633baad 41fe 491c a563 517d726028c6 06

This is a graph displaying the percentage of patients who experienced an improvement in pain from baseline (BOCF) after taking either a placebo or Durene in doses of 60mg or 120mg once a day. The horizontal axis shows the percentage of improvement in pain, while the vertical axis includes the medicines and the scale of the graph. There are no non-English characters.*
d633baad 41fe 491c a563 517d726028c6 08

This appears to be a graph showing the improvement in pain from baseline for patients taking a placebo and patients taking a medication called DULO at 60/120 mg once daily. The graph shows that the percentage of patients improved by a certain percentage, but the specific improvement percentage is cut off and unrecognizable due to the limitations of .*
d633baad 41fe 491c a563 517d726028c6 09
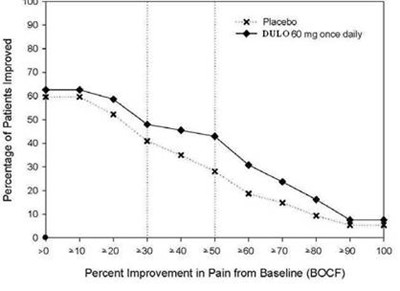
This appears to be a graph showing the percentage of patients who experienced improvement in pain after taking a medication called "Placeto" at different time intervals. The medication is taken once daily and the graph shows the percentage of improvement in pain compared to the baseline (BOCF). The x-axis shows the different time intervals and the y-axis shows the percentage of improvement in pain. However, the text is not entirely readable and some information may be missing, so this description is not 100% certain.*
d633baad 41fe 491c a563 517d726028c6 10
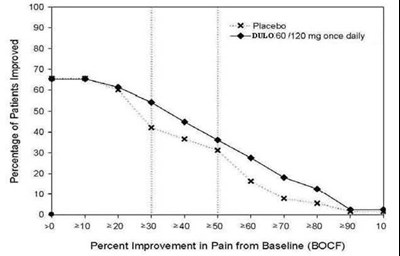
This is a graph or table displaying the percentage of patients who experienced improvement in pain intensity. The improvement rate is shown for both placebo and DULO treatments, with doses of 60 and 120 mg taken once daily. The x-axis represents the level of improvement, with values ranging from 50% to 100%. The y-axis shows the range of doses. No further information is available to provide additional context for this data.*
d633baad 41fe 491c a563 517d726028c6 11

This is the label of a medication bottle containing 30 capsules of Duloxetine hydrochloride USP. Each capsule has 22.4 mg of the active ingredient, which is equivalent to 20 mg of duloxetine. The medication must be stored in a tightly closed container at 25°C. The label includes the medication code number and the manufacturer's information.*
d633baad 41fe 491c a563 517d726028c6 13

This is a description of a medication in capsule form containing 67.3 mg of duloxetine hydrochloride USP. Each capsule is equivalent to 60 mg of duloxetine. The medication is manufactured by Inventia Healthcare Limited and comes in a bottle with 30 capsules. The recommended dosage can be found in the accompanying literature; it is advised to not use the medication if the inner seal is missing or broken. The capsules are delayed-release and should be stored in a tightly closed container at 25°C. A medication code and Aok Code are also included. There is an unvarnished area on the packaging measuring 45 x 15 mm.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.









