FDA Label for Tork Premium Hand Sanitizer Foam Alcohol-free
View Indications, Usage & Precautions
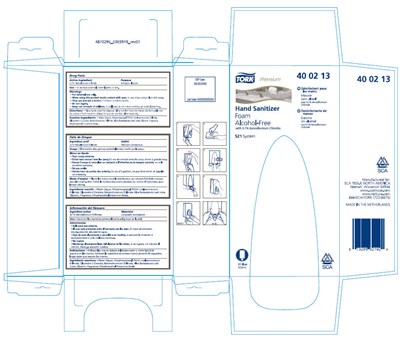
Tork Premium Hand Sanitizer Foam Alcohol-free Product Label
The following document was submitted to the FDA by the labeler of this product Sca Tissue North America. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
0.1% Benzalkonium Chloride
Purpose
Antiseptic cleanser
Use
- To decrease potentially harmful germs on skin.
Warnings
- For external use only.
- When using this product avoid contact with eyes, in case of eye contact, flush with water.
- Stop use and ask a doctor, if irritation or redness persist.
- Do not ingest.
- Keep out of reach of children, if swallowed, do not induce vomiting, get medical attention.
Directions
- Place hands under the dispenser allow 0.4ml of foam into hands, rub thoroughly over the surface of both hands for at least 30 seconds, and then allow hands dry.
Inactive Ingredients
- Water (Aqua), Dihydroxypropyl PEG-5 Linoleammonium Chloride, Glycereth- 2 Cocoate, Behentrimonium Chloride, Aloe Barbadensis Leaf Juice, Glycerin, Fragrance, Dihydroxyethyl Cocamine Oxide.
Principal Display Panel - 800 Ml Bag Carton
TORK®
Premium
40 02 13
Hand Sanitizer
Foam
Alcohol-Free
with 0.1% Benzalkonium Chloride
S21 System
FR Déinfectant pour
les mains
Mousse
Sans alcool
avec 0.1% Benzalkonium
Chloride
ES Desinfectante de
monos
Espuma
Sin alcohol
con 0.1% Benzalkonium
Chloride
27 fl oz
800ml
SCA
* Please review the disclaimer below.