Product Images Benlysta
View Photos of Packaging, Labels & Appearance
- Figure 1 - benlysta spl graphic 01
- Figure 2 - benlysta spl graphic 02
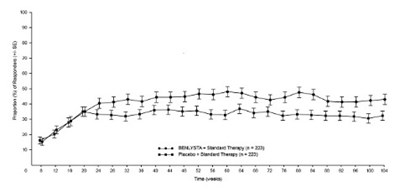
- Figure 3. Proportion (%) of SRI-4 Responders (+/- Standard Error) by Visita - benlysta spl graphic 03
- BENLYSTA prefilled syringe parts - benlysta spl graphic 04
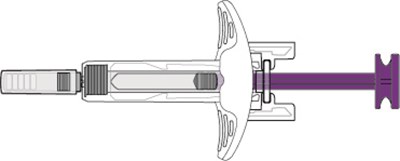
- Needle Guard - benlysta spl graphic 05
- BENLYSTA prefilled syringe - benlysta spl graphic 06
- Gauze Pad or Cotton Ball - benlysta spl graphic 07
- Supplies_Sharps_Container_PFS - benlysta spl graphic 08
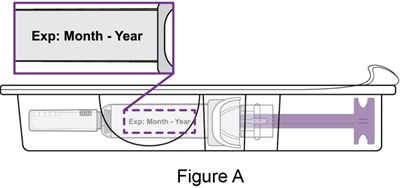
- Figure_A_PFS - benlysta spl graphic 09
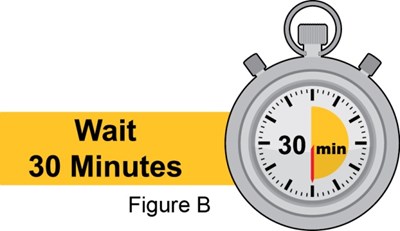
- Figure_B_PFS - benlysta spl graphic 10
- Figure_C_PFS - benlysta spl graphic 11
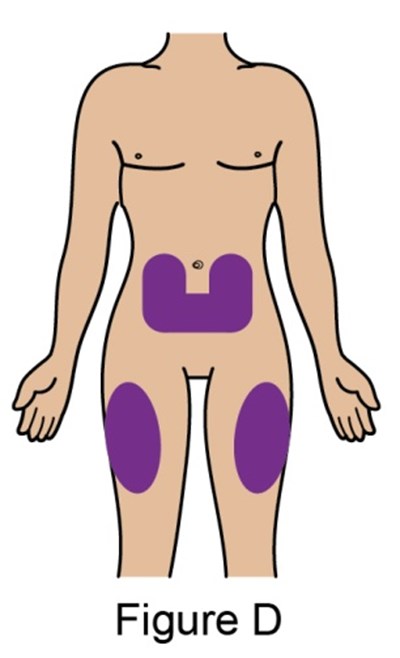
- Figure_D_PFS - benlysta spl graphic 12
- Figure_E_PFS - benlysta spl graphic 13
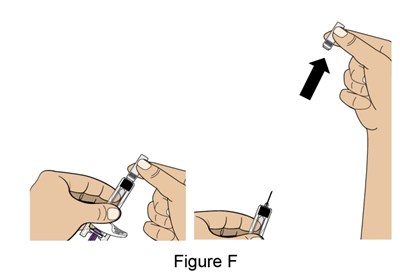
- Figure_F_PFS - benlysta spl graphic 14
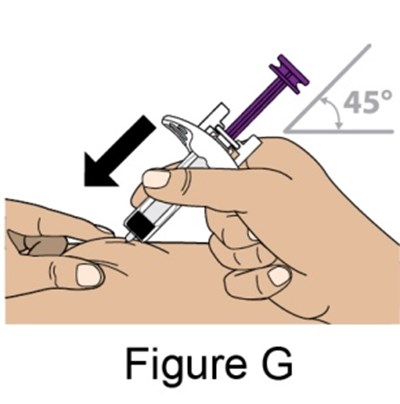
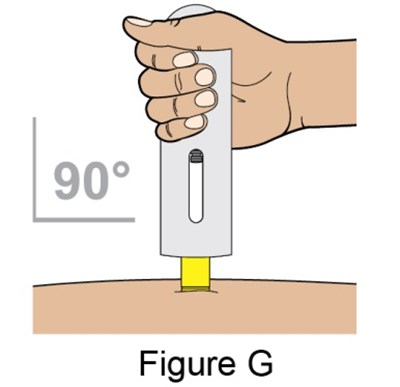
- Figure_G_PFS - benlysta spl graphic 15
- Figure_H_PFS - benlysta spl graphic 16
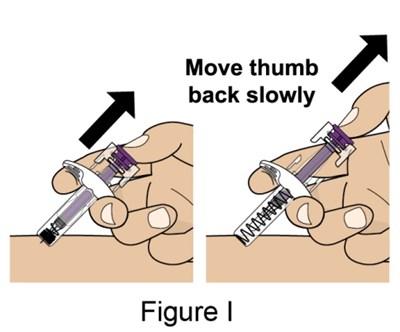
- Figure_I_PFS - benlysta spl graphic 17
- Figure_J_PFS - benlysta spl graphic 18
- Autoinjector_parts_open-view_AI - benlysta spl graphic 19
- BENLYSTA autoinjector - benlysta spl graphic 20
- Gauze Pad or Cotton Ball - benlysta spl graphic 21
- Supplies_Sharps_Container_AI - benlysta spl graphic 22
- Figure_A_AI - benlysta spl graphic 23
- Figure_B_AI - benlysta spl graphic 24
- Figure_C_AI - benlysta spl graphic 25
- Figure_D_AI - benlysta spl graphic 26
- Figure_E_AI - benlysta spl graphic 27
- Figure_F_AI - benlysta spl graphic 28
- Figure_G_AI - benlysta spl graphic 29
- Figure_H_AI - benlysta spl graphic 30
- Figure_I_AI - benlysta spl graphic 31
- Figure_J_AI - benlysta spl graphic 32
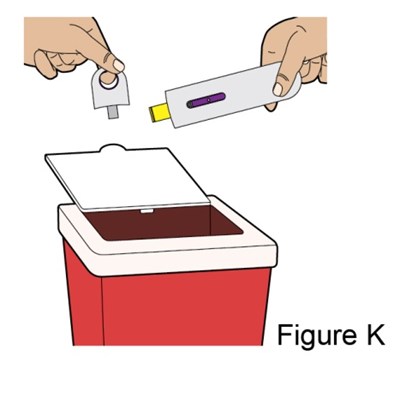
- Figure_K_AI - benlysta spl graphic 33
- benlysta 120 mg per vial carton - benlysta spl graphic 34
- benlysta 400 mg per vial carton - benlysta spl graphic 35
- Benlysta 200 mg-mL 4 count carton - benlysta spl graphic 36
Product Label Images
The following 36 images provide visual information about the product associated with Benlysta NDC 49401-088 by Glaxosmithkline Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 3. Proportion (%) of SRI-4 Responders (+/- Standard Error) by Visita - benlysta spl graphic 03

BENLYSTA prefilled syringe parts - benlysta spl graphic 04

This text is a list of words or phrases and it seems to refer to some inspection related to needles and a cap window. However, it lacks context and it is not possible to provide a more precise description with this information.*
Autoinjector_parts_open-view_AI - benlysta spl graphic 19

This appears to be a list of items related to an inspection, including a ring cap window, Coes (which may refer to a type of wrench), gold and grey items, a needle guard and stopper, and an expiration date. It's unclear what specific product or process this pertains to.*
benlysta 120 mg per vial carton - benlysta spl graphic 34

Benlysta (belimumab) is a medication available in single-dose vials for intravenous infusion after dilution with sterile water for injection, USP. It is to be further diluted before use with 0.9% Sodium Chloride Injection, USP, 0.45% Sodium Chloride Injection, USP, or Lactated Ringer’s Injection, USP. Federal law requires dispensing of Benlysta with the medication guide provided. RX only.*
benlysta 400 mg per vial carton - benlysta spl graphic 35

Benlysta (belimumab) is an injection for intravenous infusion after dilution. The concentration of Benlysta is 80 mg/mL after reconstitution with sterile water for injection, USP. A further dilution is required before use to 250 mL of either 0.9% Sodium Chloride Injection, USP or 0.45% Sodium Chloride Injection, USP or Lactated Ringer's Injection, USP. BENLYSTA must be dispensed only with the Medication Guide provided with the carton as required by Federal Law. This is a single-dose vial and any unused portion must be discarded.*
Benlysta 200 mg-mL 4 count carton - benlysta spl graphic 36

This is a description of a medication called Benlysta, which is an injection administered subcutaneously once a week. It comes in a package that includes four single-dose 1-mL profiled autoinjectors, as well as instructions for use, a medication guide, and prescribing information. Dispensing of Benlysta is required by law with the medication guide provided in the package. The package is labeled with NDC code 49401-088-35 and is only available with a prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.