Product Images Tivicay
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Tivicay NDC 49702-226 by Viiv Healthcare Company, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
tivicay spl graphic 05

This is a very brief and simple instruction telling someone to administer medicine to a child.*
tivicay spl graphic 06
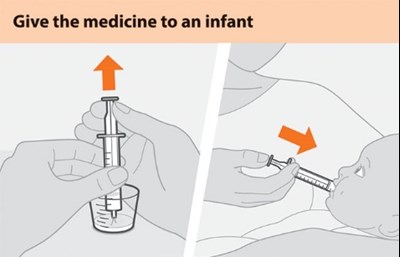
This text is a simple instruction stating to administer medicine to an infant.*
tivicay spl graphic 08

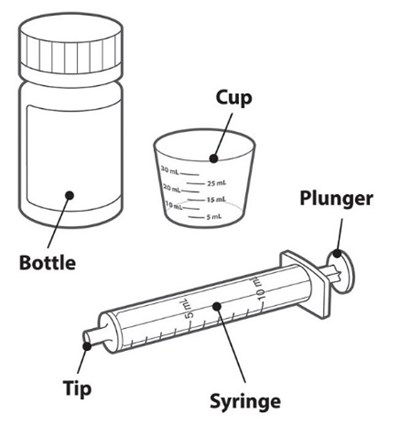
This is a prescription medication called TivicayPD with the active ingredient dolutegravir. It is available in tablet form and is not interchangeable with other medications. Each tablet contains 5mg of dolutegravir, and the bottle comes with 60 tablets and a dosing syringe and cup. The medication should not be chewed, cut, or crushed, and should not be used if the membrane seal under the cap is missing or broken. The prescribing information should be consulted for dosing instructions.*
tivicay spl graphic 09

This is a prescription drug with the name Tivicay, whose active ingredient is dolutegravir. It comes in a film-coated tablet formulation with each tablet containing 10 mg of dolutegravir in the form of dolutegravir sodium. It is packed in a bottle containing 30 tablets and should be stored at controlled room temperature. The prescribing information should be consulted for dosage recommendations. The drug is marketed by ViiV Healthcare.*
tivicay spl graphic 10

This is a medication with the name "Ivica" (dolutegravir) which comes in film-coated tablet form with each tablet containing 25mg of dolutegravir sodium. It is a prescription medication with dosage information on the prescribing information. It should be stored at a controlled room temperature of 25°C, with excursions permitted at 10-30°C (59° to 86°F). The manufacturer is located in Research Triangle Park, NC 27709, and it is made in Japan.*
tivicay spl graphic 11

This is a prescription drug called Tivicay (dolutegravir) used for treating HIV. Each film-coated tablet contains dolutegravir sodium equivalent to 50 mg of dolutegravir. It comes in a bottle of 30 tablets and should be stored at a controlled room temperature of 25°C (77°F), with excursions permitted to 15°t0 30°C (59° to 86°F). The manufacturer is ViiV Healthcare based in Research Triangle Park, NC, and the medication is made in Japan. For dosage information, please refer to the prescribing information.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.