Product Images Apretude
View Photos of Packaging, Labels & Appearance
- structural formula of cabotegravir - apretude spl graphic 01
- Figure 1. Cumulative Incidence of HIV-1 Infections in HPTN 083 - apretude spl graphic 02
- Figure 2. Cumulative Incidence of HIV-1 Infections in HPTN 084 - apretude spl graphic 03
- Instructions for Use header - apretude spl graphic 04
- storage information - apretude spl graphic 05
- Kit contents - apretude spl graphic 06
- Figure A - apretude spl graphic 07
- Figure B - apretude spl graphic 08
- Figure C - apretude spl graphic 09
- Figure D - apretude spl graphic 10
- Figure E - apretude spl graphic 11
- Figure F - apretude spl graphic 12
- Figure G - apretude spl graphic 13
- Figure H - apretude spl graphic 14
- Figure I - apretude spl graphic 15
- Figure J - apretude spl graphic 16
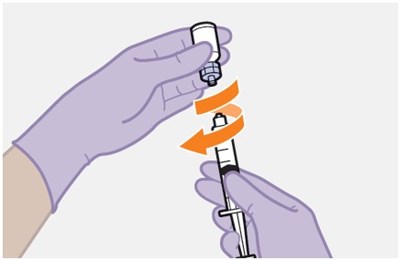
- Figure K - apretude spl graphic 17
- Figure L - apretude spl graphic 18
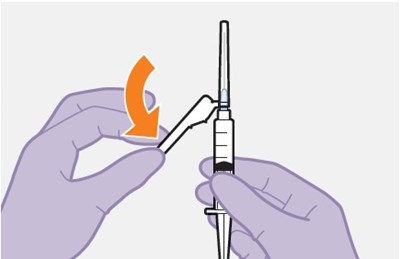
- Figure M - apretude spl graphic 19
- Figure N - apretude spl graphic 20
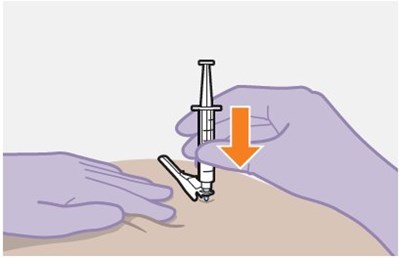
- Figure O - apretude spl graphic 21
- Figure P - apretude spl graphic 22
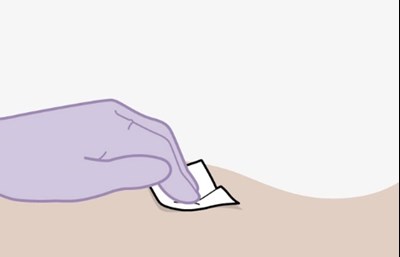
- Figure Q - apretude spl graphic 23
- Figure R - apretude spl graphic 24
- Figure S - apretude spl graphic 25
- Apretude 3 mL kit carton - apretude spl graphic 26
Product Label Images
The following 26 images provide visual information about the product associated with Apretude NDC 49702-264 by Viiv Healthcare Company, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Instructions for Use header - apretude spl graphic 04

INSTRUCTIONS FOR USE: "Pretude" is an extended-release injectable suspension with 600 mg/3 mL dosage (200 mg/mL). It is intended for intramuscular use only in the gluteal region of the body.*
Kit contents - apretude spl graphic 06
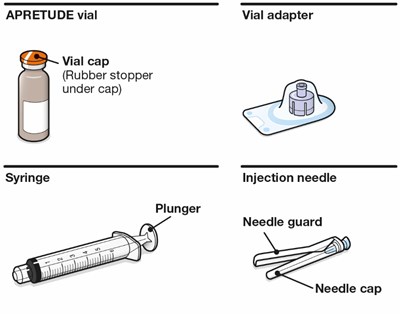
The text describes a medical device called "APRETUDE vial Vial adapter" that includes a vial cap with a rubber stopper underneath. The device also involves a syringe with an injection needle, a plunger, a needle guard, and a needle cap.*
Figure A - apretude spl graphic 07

This text is a simple reminder to check the expiration date of medicine before use.*
Apretude 3 mL kit carton - apretude spl graphic 26

Apretude is a prescription medication for healthcare professional administration only. It is an injectable suspension containing 600 mg/3 mL cabotegravir extended-release. It should only be administered into the gluteal muscle, using the vial adapter, syringe, and 23 gauge, 1% inch injection needle provided. Prescribing information, patient information, and instructions for use are also included. The National Drug Code for this medication is 49702-264-23.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.