FDA Label for Medactive Patient Friendly Stannous Fluoride Rinse
View Indications, Usage & Precautions
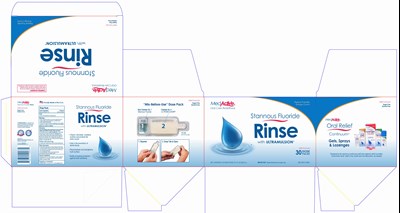
Medactive Patient Friendly Stannous Fluoride Rinse Product Label
The following document was submitted to the FDA by the labeler of this product Integrate Oral Care, Llc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Active Ingredient
Stannous Fluoride 0.1%
Indications & Usage
Uses
Aids in the prevention of dental decay.
Warnings
Warnings
- When using this product, do not use longer than four weeks unless recommended by a medical doctor and/or dental professional.
- If more than used for rinsing is swallowed, seek medical attention or contact a Poison Control Center immediately.
- This product may produce surface staining of the teeth. Adequate toothbrushing may prevent these stains which are not harmful or permanent and may be removed by your dental professional.
- Do not refrigerate. Store at room temperature.
- See Rinse or box for expiration date.
Other Information
Dosage & Administration
Directions
Use immediately after preparing the rinse as follows:
Adults and children 6 years or age and older:
- Place both thumbs on Blue Chamber No. 1 and Press down firmly to squeeze fluoride into Chamber No. 2.
- Gently Shake.
- Snap Tab to Open by bending at line on the cap. Dispense half of mixture into the mouth and Rinse for 30 seconds. Spit.
- Gargle with remainder of mixture for 30 seconds. Spit.
- Avoid any food or drinks for at least 15 minutes after use.
- Consult your medical doctor and/or dental professional.
Children under 6 years of age:
Inactive Ingredient
Inactive Ingredients
dimethocone, EDTA, FD&C Green No. 3, flavor, glycerin, poloxamer 407, potassium sorbate, sodium saccharin, sorbital, sucralose, water
Otc - Questions
Questions or comments?
1-866-887-4867 M-F 9 am - 5 pm EST or www.integrateoralcare.com
Otc - Keep Out Of Reach Of Children
Keep out of reach of children.
Otc - Purpose
Purpose
Cavity Protection
Prinicpal Display Panel - 30 Dose Packs
MedActive
Oral Care Redefined
Patient-Friendly
Orange Crème
Stannous Fluoride Rinse with ULTRAMULSION
Mild Formula
30 Dose Packs
NET CONTENTS: 30 DOSE PACKS 31.2 FL OZ (923 mL)
IMPORTANT: Read directions for proper use.
NDC 49741-1005-3
Principal Display Panel - 10 Dose Packs
MedActive
Oral Care Redefined
Patient-Friendly
Orange Crème
Stannous Fluoride Rinse with ULTRAMULSION
Mild Formula
10 Dose Packs
NET CONTENTS: 10 DOSE PACKS 10.4 FL OZ (307 mL)
IMPORTANT: Read directions for proper use.
NDC 49741-1006-2
* Please review the disclaimer below.