Product Images Amlodipine Besylate And Benazepril Hydrochloride
View Photos of Packaging, Labels & Appearance
- This is the 10 mg/20 mg label - 10 mg 20 mg100
- This is the 10 mg/40 mg label - 10 mg 40 mg100
- This is 2.5 mg/10 mg label - 2.5mg 10mg100
- This is the 5 mg/10 mg label - 5mg 10mg100
- This is the 5 mg/20 mg label - 5mg 20 mg100
- This is 5 mg/40 mg label - 5mg 40 mg100
- This is the benazepril structure - structure1
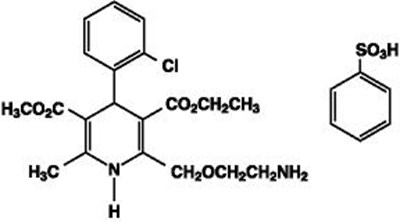
- This is amlodipine/besylate structure - structure2
Product Label Images
The following 8 images provide visual information about the product associated with Amlodipine Besylate And Benazepril Hydrochloride NDC 49884-952 by Endo Usa, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
This is the 10 mg/20 mg label - 10 mg 20 mg100

This is a prescription drug with the National Drug Code (NDC) of 49884-932-01. The medication is called Amlodipine Besylate and Benazepril Hydrochloride Capsules. Each capsule contains 10 mg of amlodipine and 20 mg of benazepril hydrochloride. The dosage should be followed as described in the package insert. The drug should be stored at 20° to 25°C (68° to 77°F) and kept away from moisture. This medication should be kept out of reach of children and be dispensed in a tightly sealed container. An information leaflet is provided separately to each patient, and the controlling number is 3 49884-932-015.*
This is the 10 mg/40 mg label - 10 mg 40 mg100

This is a description of a medication with the NDC code 49884-953-01. The medication contains Amlodipine Besylate and Benazepril Hydrochloride and is provided in separate capsules to each patient. Each capsule contains 10 mg of amlodipine besylate and 40 mg of benazepril hydrochloride. Dosage instructions can be found in the package insert. The medication should be kept out of reach of children and stored in a tight container, protected from moisture, at a temperature between 20° to 25°C (68° to 77°F). The document also displays a control number and an expiration date.*
This is 2.5 mg/10 mg label - 2.5mg 10mg100

This is a package insert for Amlodipine Besylate and Benazepril Hydrochloride Capsules. The capsules are available in strengths of 2.5mg/10mg and come in a package of 100 capsules. The drugs should be kept out of reach of children, dispensed by a pharmacist in a tight container, and stored at a temperature between 20° to 25°C. The text also provides manufacturer and product code numbers.*
This is the 5 mg/10 mg label - 5mg 10mg100

These are Amlodipine Besylate and Benazepril Hydrochloride Capsules with a dosage of 5 mg/10 mg, which are available in a packet of 100 Capsules. Each Capsule contains 5 mg of Amlodipine Besylate and 10 mg of Benazepril Hydrochloride. It is a prescription drug that should be kept out of the reach of children and should be stored in a tight container between 20°C to 25°C. It is manufactured in Spring Valley, NY 10977. Please refer to the package insert for dosage guidelines.*
This is the 5 mg/20 mg label - 5mg 20 mg100

This is a description of a medication called Amlodipine Besylate and Benazepril Hydrochloride Capsules. It is available in a bottle containing 100 individual capsules with each capsule containing 5mg of amlodipine besylate and 20mg of benazepril hydrochloride. The recommended dosage for this medication is not given and it should be kept out of reach of children in a tight container. The medication should be stored at a temperature between 20 and 25°C and protected from moisture. The name and location of the manufacturing company, Par Pharmaceutical Companies, Inc., are also provided along with a control number and expiration date.*
This is 5 mg/40 mg label - 5mg 40 mg100

This is a medication in capsule form with the NDC code 49884-952-01. Each capsule contains amlodipine besylate equivalent to 5 mg amlodipine and 40 mg benazepril hydrochloride. It is recommended to read the package insert and store the drugs in a tight container at temperatures ranging from 20 to 25°C (68 to 77°F). The medication is provided by Par Pharmaceutical Companies, Inc., in Spring Valley, NY 10977. No further information is available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.