FDA Label for Terbinafine Hydrochloride
View Indications, Usage & Precautions
Terbinafine Hydrochloride Product Label
The following document was submitted to the FDA by the labeler of this product A-s Medication Solutions. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Other
Drug Facts
Distributed by: Taro Pharmaceuticals U.S.A., Inc.
Hawthorne, NY 10532
Active Ingredient
Terbinafine hydrochloride 1%
Purpose
Antifungal
Uses
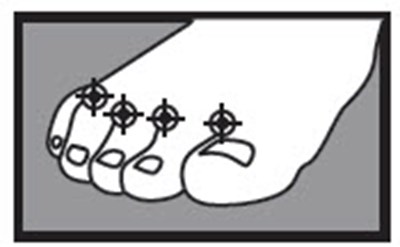
- cures most athlete's foot (tinea pedis)
- cures most jock itch (tinea cruris) and ringworm (tinea corporis)
- relieves itching, burning, cracking and scaling which accompany these conditions
Warnings
For external use only
Do Not Use
- on nails or scalp
- in or near the mouth or eyes
- for vaginal yeast infections
Otc - When Using
When using this product do not get into eyes. If eye contact occurs, rinse thoroughly with water.
Otc - Stop Use
Stop use and ask a doctor if too much irritation occurs or gets worse
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
| 1 week between the toes 2 weeks on the bottom or sides of the foot |
Other Information
TAMPER EVIDENT: DO NOT USE IF THE SEAL ON THE TUBE IS PUNCTURED OR NOT VISIBLE.
- store at controlled room temperature 20° to 25°C (68° to 77°F)
- see carton or tube crimp for lot number and expiration date
Inactive Ingredients
benzyl alcohol, cetyl alcohol, cetyl palmitate, isopropyl myristate, polysorbate 60, purified water, sodium hydroxide, sorbitan monostearate, stearyl alcohol
Questions?
call 1-866-923-4914
How Supplied
Product: 50090-1278
NDC: 50090-1278-0 15 g in a TUBE / 1 in a CARTON
* Please review the disclaimer below.