Product Images Drospirenone And Ethinyl Estradiol
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Drospirenone And Ethinyl Estradiol NDC 50090-2494 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
cid:[email protected] - 6D85F252 307C 4AA8 9572 1E74C9819283 00

cid:[email protected] - 6D85F252 307C 4AA8 9572 1E74C9819283 01

figure1 - figure1
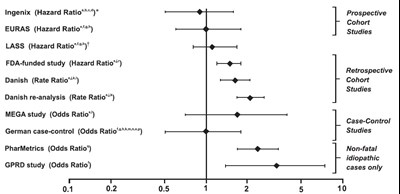
This text seems to be describing different hazard, rate, and odds ratio studies conducted in different countries and funded by different organizations. It also mentions the types of studies conducted, such as prospective and retrospective cohort studies, case-control studies, and non-fatal idiopathic cases only. The numbers at the bottom may indicate some sort of numerical data. Overall, this text seems to be a list or summary of different medical studies and their respective ratios and study types.*
figure2 - figure2

This is a figure showing the likelihood of developing a blood clot (VTE) for non-pregnant COC (contraceptive pill) users and non-users, pregnant women and postpartum women (up to 12 weeks). The number of women with a blood clot per 10,000 women years is shown on the y-axis. The background text provides additional information on the ranges of likelihood for each group. The pregnancy data is based on the actual duration of pregnancy in the reference studies and also mentions an assumed rate for pregnancy durations of 9 months.*
drospirenone structure - image 03

Drospirenone is a medication used in birth control pills and hormone replacement therapy. It is a synthetic progestin that works by preventing ovulation and thickening cervical mucus to prevent sperm from reaching an egg.*
img1 - img1

This text is a chart showing the effectiveness of different methods of birth control in preventing pregnancy. It includes the rates of pregnancies per 100 women in one year for various methods, such as hormonal implants, injections, and devices, as well as non-hormonal options like the diaphragm, spermicide, and withdrawal. It also suggests the effectiveness of abstaining from sex during the most fertile days of the monthly cycle.*
img3 - img3

This is a table that lists the ranges of blood clot occurrences in non-pregnant women and non-COC users, as well as ranges for pregnancy and postpartum (up to 12 weeks only). The table shows the number of women with blood clots out of 10,000 women years (WY). The pregnancy data is based on the actual duration of pregnancy in reference studies, but a model assumption that pregnancy duration is nine months gives a rate of 7-27 per 10,000 WY.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


