Product Images Farxiga
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Farxiga NDC 50090-3482 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 4 - Figure 4
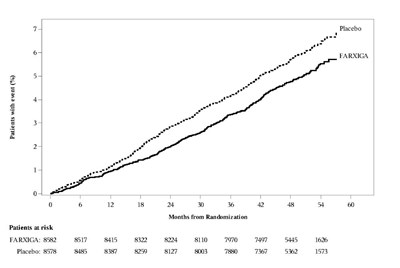
This is a table showing the number of patients who had an event and the number of patients at risk for a clinical trial involving FARXIGA and placebo. The table lists the months from randomization and the patient count for each month.*
Figure 6A - Figure 6a
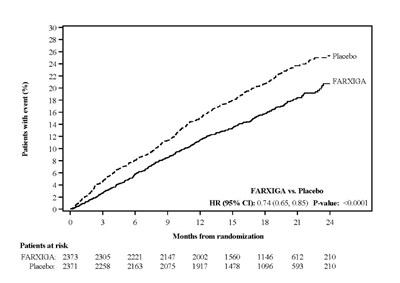
This is a clinical study result for the drug FARXIGA compared to a Placebo for the treatment of an undisclosed condition. The study followed patients for 24 months and shows a reduction in the occurrence of the event in the group taking FARXIGA compared to the Placebo group.*
Figure 6B - Figure 6b

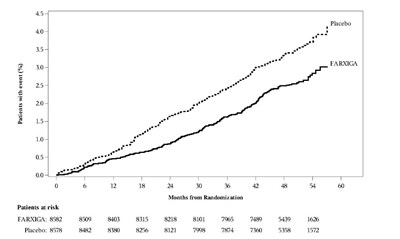
This is a clinical trial comparison between the drug FARXIGA and a placebo. The graph displays the percentage of patients with a medical event over a two-year period starting from randomization. Patients taking FARXIGA had a lower rate of medical events - the hazard ratio was 0.82 (with a 95% confidence interval of 0.69 to 0.98) and a P-value of 0.0294. The number of patients at risk over the 24 months is shown for both treatment groups.*
Figure 6C - Figure 6c
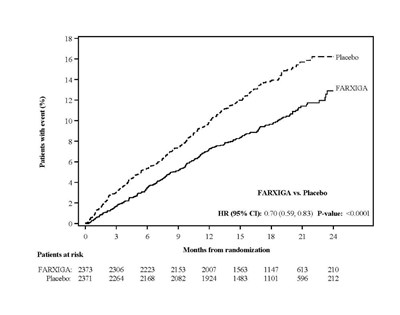
This text appears to be a comparison of the effectiveness of the drug FARXIGA versus a placebo, with data on the number of patients with an event (%) over a period of 24 months, along with hazard ratios and p-values. The data is presented in a table format with patients at risk for each month.*
Figure 1 - farxiga fig1
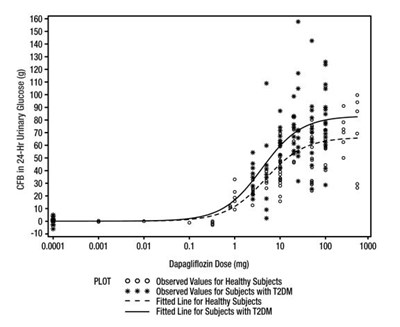
This appears to be a graph that shows the correlation between the dosage of a medication called Dapagiiizin and the amount of glucose present in the urine of study participants. There are two separate groups represented on the graph: healthy subjects and those with a condition abbreviated to "T20M". The graph includes two fitted lines to represent the correlation between dosage and glucose levels for these groups.*
Figure 8 - figure 8
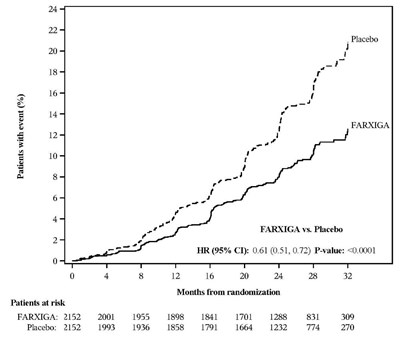
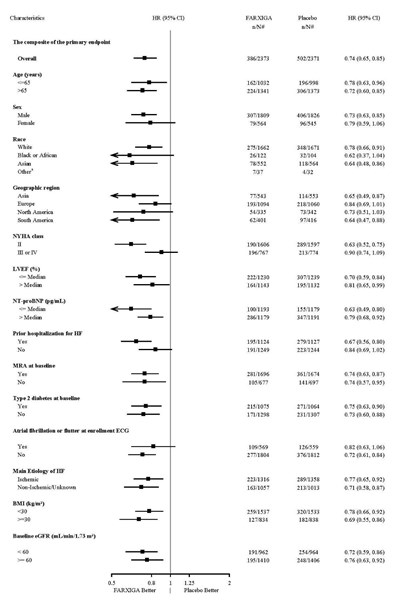
This is a statistical report showing a comparison between Farxiga and a Placebo. It includes data on the number of patients with an event, percentage, Hazard Ratio, P-value, and patients at risk. The report also includes a chart with months from randomization and patients at risk over time.*
FARXIGA Figure 3 - image 02
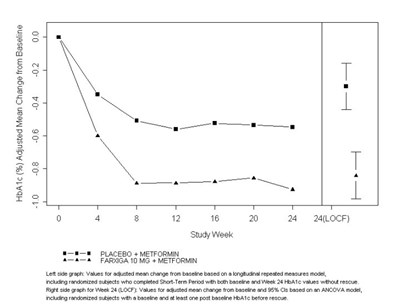
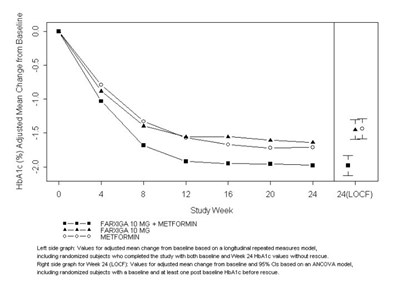
This is a graph showing the adjusted mean change from baseline HbA1c levels over time in a study with randomized subjects who completed a short-term period without rescue. The graph includes two lines, one for the placebo plus metformin group and the other for the Farxiga 10 mg plus metformin group. The graph also includes error bars and study week intervals. The text also includes a description of the statistical models used to generate the data.*
Label Image - lbl500903481

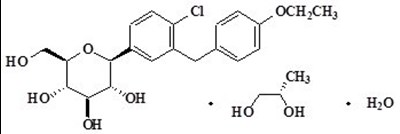
This is a medication product description for Dapagliflozin 16mg tablets. The recommended storage for the tablets is at 68-77 degrees Fahrenheit. The product code is EI57-0 and the package includes 30 tablets.*
Label Image - lbl500903482

Product No. 6796-0 is a product identified as "LoT' FARKIGA" which contains the active ingredient Dapagliflozin. Further information about the product is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.