Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Donepezil Hydrochloride NDC 50090-3537 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - 41d7651e b91a 4118 8719 bf2f663d82a2 02

This appears to be a table or graph showing the ADAS-cog change from baseline and clinical improvement for three different drug treatments over the course of several weeks. Without more context or information, it is difficult to provide a more detailed description.*
Figure 2 - 41d7651e b91a 4118 8719 bf2f663d82a2 03

The text refers to some kind of graph or data visualization related to the "Cumulative Percentage of Patents". It also mentions changes in the "ADAS-cog" score between a study group and a placebo group. There are percentages listed for each group. The text also includes numerical values (presumably related to a chart/graph) for two additional groups: "Smoiday" and "omgiy". Finally, it lists two doses of a drug called "Donepezil hydrochloride" (10 mg/day and 5 mg/day).*
Figure 3 - 41d7651e b91a 4118 8719 bf2f663d82a2 04

This text presents a table showing the percentage of patients who experienced improvement, worsening, or no change when using Donepezil hydrochloride and placebo for treatment. The table also includes the CIBIC-plus Rating.*
Figure 4 - 41d7651e b91a 4118 8719 bf2f663d82a2 05

This appears to be a chart showing the ADAS-cog change from baseline for three different treatments: donepezil hydrochloride 10 mg/day, donepezil hydrochloride 5 mg/day, and placebo. The chart measures the mean change in ADAS-cog score after weeks of drug treatment. The symbol "+" with the value "5E" is also present, but the meaning is not clear.*
Figure 5 - 41d7651e b91a 4118 8719 bf2f663d82a2 06

This is a table detailing the percentage of patients and their response to various dosages of Donepezil hydrochloride and Placebo drugs in terms of their change in ADAS-cog score. There is also a mention of Chsnne foms Buselin, but the context is unclear.*
Figure 6 - 41d7651e b91a 4118 8719 bf2f663d82a2 07

This is a table presenting the percentage of patients for the drugs Aricept 10mg/day, Aricept 5mg/day and placebo. The table shows the CIBIC-plus rating for the patients as Markedly Improved, Moderately Improved, Minimally Improved, No Change, Minimal Worse, Moderately Worse and Markedly Worse.*
Figure 8 - 41d7651e b91a 4118 8719 bf2f663d82a2 09

The text provided is a pharmaceutical prescription, indicating the medication "Donepezil Hydrochloride" with a recommended dosage of "10mg/day". The text also includes a mention of the control group "Placebo", and some unidentifiable characters. The mention of "Baseline" suggests that this may be a clinical trial or a medical study.*
Label Image - lbl500903537

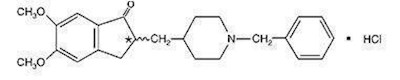
Product: Donepezil HCL 56, 90 Tablets. Store at 5970 8 Degrees Celsius. This could be a medication used to treat dementia or Alzheimer's disease.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.