Product Images Tadalafil
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Tadalafil NDC 50090-4276 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
tadalafil-fig1 - tadalafil fig1
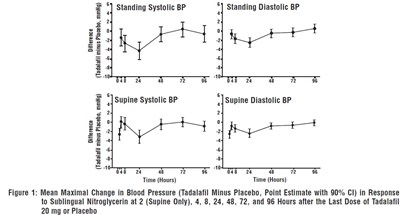
The text appears to be describing a graph (Figure 1), which shows the mean change in blood pressure in response to sublingual nitroglycerin at various time intervals after taking either Tadalafil or a placebo. The graph displays the difference between standing systolic blood pressure, standing diastolic blood pressure, supine systolic blood pressure, and supine diastolic blood pressure. The point estimate with a 90% confidence interval is also shown.*
tadalafil-fig2 - tadalafil fig2

This is a description of a study that involves the administration of Tadalafil and Doxazosin, with the dosage of Tadalafil being 20 mg and the dosage of Doxazosin being 8 mg. A placebo and Doxazosin at 8 mg were also administered. The study measured the mean change in systolic blood pressure over time after the administration of Doxazosin.*
tadalafil-fig3 - tadalafil fig3

This is a report from a study involving the use of 20mg of tadalafil and 8mg of doxazosin at different times throughout the day. The study measures the mean change from a time-matched baseline in systolic blood pressure. The text is not available for the figure produced.*
tadalafil-fig4 - tadalafil fig4
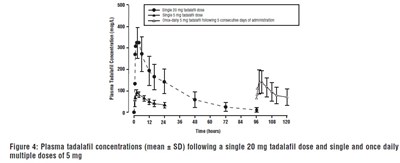
Plasma concentration of Tadalafil (a medication used to treat erectile dysfunction) is shown in this figure. The graph illustrates the concentration levels after administering a single dose of 20mg and once daily multiple doses of 5mg for five consecutive days. The x-axis displays the time in hours while the y-axis represents the concentration in mg/L. Error bars are shown for each data point to indicate variability.*
tadalafil-fig5.jpg - tadalafil fig5
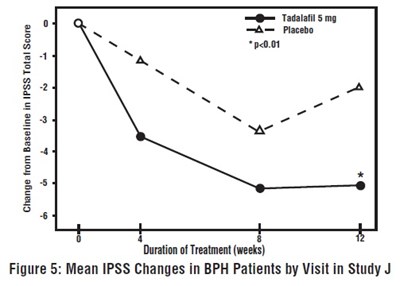
This is a figure from Study J which compares the mean changes in IPSS total score in BPH patients by visit. The treatment duration in weeks is also shown on the X-axis. The medications being compared are Tatalafil 5 mg and placebo. No further information is available.*
tadalafil-fig7.jpg - tadalafil fig7

The text describes a figure (Figure 7) showing the mean total IPSS changes by visit in patients with BPH taking Tadalafil and Finasteride for once-daily use. The figure shows the weeks of therapy along with the LS mean change from baseline, and indicates that the treatment difference is significant (p<0.001).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



