Product Images Alendronate Sodium
View Photos of Packaging, Labels & Appearance
- 1203dd0c 809a 4409 b948 b5e816b8a63a 01
- 1203dd0c 809a 4409 b948 b5e816b8a63a 02
- 1203dd0c 809a 4409 b948 b5e816b8a63a 03
- 1203dd0c 809a 4409 b948 b5e816b8a63a 04
- 1203dd0c 809a 4409 b948 b5e816b8a63a 05
- 1203dd0c 809a 4409 b948 b5e816b8a63a 06
- 1203dd0c 809a 4409 b948 b5e816b8a63a 07
- Label Image - lbl500904692
Product Label Images
The following 8 images provide visual information about the product associated with Alendronate Sodium NDC 50090-4692 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
1203dd0c 809a 4409 b948 b5e816b8a63a 03

This text provides information on the results of an osteoporosis treatment study in postmenopausal women. Specifically, it shows an increase in bone mineral density (BMD) after three years of treatment with Alendronate Sodium 10 mg/day. The table shows BMD measurements for different skeletal sites, including the lumbar spine, femoral neck, and trochanter.*
1203dd0c 809a 4409 b948 b5e816b8a63a 04

Osteoporosis Treatment Studies in Postmenopausal women comparing the effect of taking Alendronate Sodium 10 mg/day versus placebo over time. The study measured the percent change in Bone Mineral Density (BMD) of the Lumbar Spine from the starting point. The study was conducted in both the US and multiple countries. The provided data shows a graph with axes labeled for time in months and BMD change from baseline in percentage. The graph consists of multiple lines representing different data points. No further information is available to provide more context.*
1203dd0c 809a 4409 b948 b5e816b8a63a 05

This appears to be a table with headings and subheadings related to studies on bone mineral density in postmenopausal women. Specifically, it shows data on the percentage change in BMD from baseline in two and three-year studies, with separate information on the lumbar spine, femoral neck, and total body. It also includes information on sodium intake.*
1203dd0c 809a 4409 b948 b5e816b8a63a 07
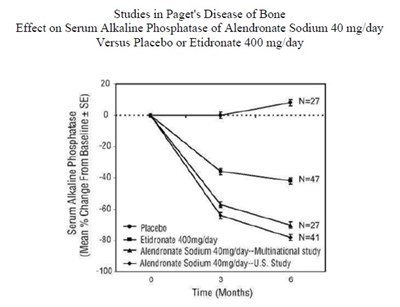
A study comparing the effects of Alendronate Sodium 40mg/day versus Placebo or Etidronate 400mg/day on serum alkaline phosphatase in Paget's disease of bone. The study included a total of 27 participants and displayed the serum levels of alkaline phosphatase over time (in months) for each treatment. Mean percentage changes from baseline were also reported. No further details were available for review.*
Label Image - lbl500904692

This appears to be a label or tag for a medication package containing 4 tablets in a blister pack. It lists a product code as "irmu:'z No. 6050-0" and instructions to store at a location with the address "6810 7 OkGAEES."*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


