Product Images Alendronate Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Alendronate Sodium NDC 50090-5677 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
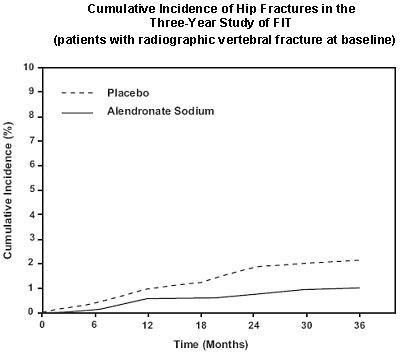
Figure 2 - alendronate fig2

This text provides the results of studies conducted on the effects of Alendronate 10 mg/day in the treatment of postmenopausal women with osteoporosis. The results are presented in the form of the mean percentage increase of Bone Mineral Density (BMD) relative to placebo, along with standard error (SE), values for the lumbar spine, femoral neck, and trochanter regions. The BMD increased by 12% in three years.*
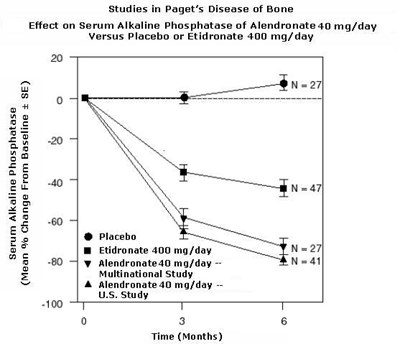
Figure 3 - alendronate fig3

This is a graph showing the effect of Alendronate 10 mg/day versus a placebo on Lumbar Spine BMD Percent Change From Baseline in postmenopausal women with Osteoporosis. The study was a multinational one and lasted for 36 months. The graph shows that there was a significant increase in BMD percentage for the group that was treated with Alendronate 10 mg/day compared to the placebo group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.