Product Images Divalproex Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Divalproex Sodium NDC 50090-5843 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - divalproex fig1

The text provides a Figure 1 with two study groups named "Study 1" and "Study 2", which were evaluated based on years (YRS) and Wisconsin Wiener Scale (WIRS) respectively. The text also mentions some abbreviations such as PBO, DVPX and DVPK P80, and indicates that the statistical significance for the results is less than 0.05. DVPX is associated with divalproex sodium delayed-release tablets.*
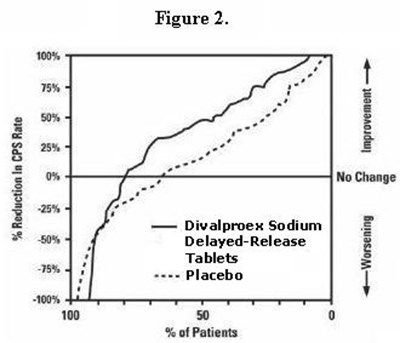
Figure 3 - divalproex fig3

This text represents a graph or chart labelled "Figure 3" which shows the percentage reduction in CPS (Chronic Prostatitis Symptom) rate. The graph displays two categories "High Dose" and "Low Dose" with percentage of patients on the Y-axis and "Improvement", "No change", and "Worsening" on the X-axis. It indicates the percentage of patients showing improvement, no change and worsening.*
Figure 4 - divalproex fig4

This text provides a comparison of mean 4-week migraine rates between Placebo and Divalproex Sodium Delayed-Release Tablets in Study 1 and Study 2. It also includes information on the mean dose of divalproex sodium delayed-release tablets used in the studies.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.