Product Images Budesonide And Formoterol Fumarate Dihydrate
View Photos of Packaging, Labels & Appearance
- Counter Image 15 - 308708 rapihaler counter 15 (black;130;485)
- Counter Image 80 - 308710 rapihaler counter 80 (black;130;485)
- Figure 1 - figure1bodytext
- Figure 2 - figure2bodytext
- Figure 3 - figure3bodytext
- Figure 4 - figure4bodytext
- Figure 5 - figure5bodytext
- Figure 6 - figure6bodytext
- Figure 7 - figure7bodytext
- budesonide structural formula - image 01
- structural fumarate - image 02
- Figure 1 - image 03
- Figure 2 gray arrows - image 04
- Figure 3 - image 05
- Figure 4 - image 06
- Figure 5 - image 07
- Label Image - lbl500906327
Product Label Images
The following 17 images provide visual information about the product associated with Budesonide And Formoterol Fumarate Dihydrate NDC 50090-6327 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - figure1bodytext
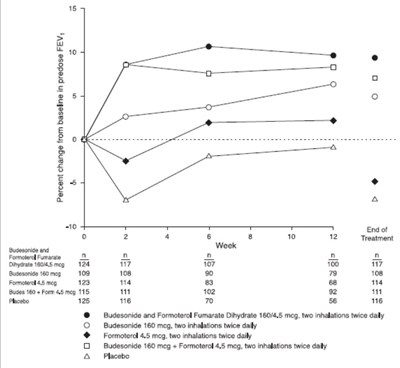
This looks like a table showing results of a treatment involving Budesonide and Formoterol Fumarate Dihydrate. The table shows different dosages and inhalations and their corresponding effectiveness. The treatment may be used for respiratory conditions.*
Figure 3 - figure3bodytext
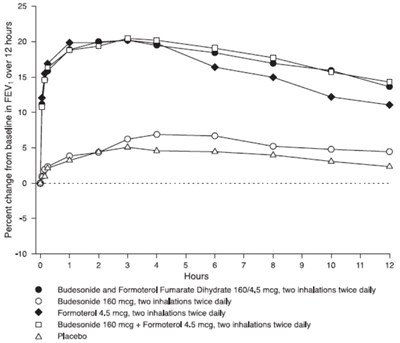
Not available. The text appears to be a combination of various characters and symbols that do not form coherent sentences or meaningful information.*
Figure 4 - figure4bodytext
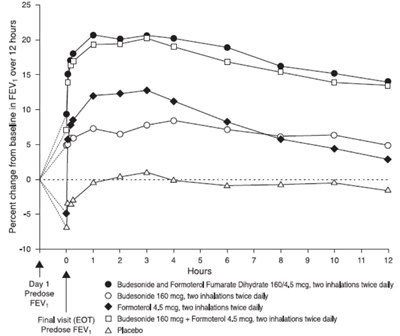
This is a graph showing the percent change from baseline in FEV over 12 hours. The x-axis represents the hours, and the y-axis represents the percent change in FEV. The graph has four lines, one labeled Day 1 with Budesonide and Formoterol Fumarate Dihydrate, another labeled Predose O Budesonide, a third labeled FEVY with Formoterol, and the final labeled Final visit (EOT) with Budesonide and Formoterol. The graph also includes a legend explaining the different lines. There is no available information on what study this graph is from or what it represents.*
Figure 6 - figure6bodytext
This is a clinical study report for the drug combination of Budesonide and Formoterol Fumarate Dihydrate for inhalation. It includes the measurement of percentage change from baseline in pre-dose FEV. Different dosages of the drug combination, as well as individual dosages of Budesonide and Formoterol, are compared against a placebo. The report includes numerical data and abbreviations for relevant terms.*
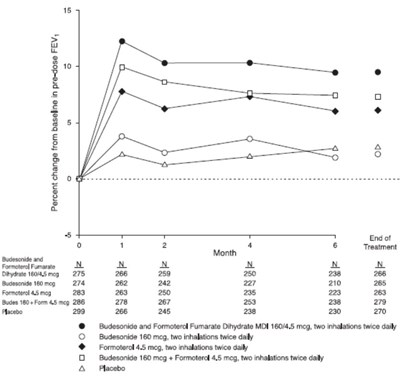
Figure 7 - figure7bodytext

This appears to be a table of data related to the percent change in FEV (forced expiratory volume) from baseline in a study of different treatments for what is likely an obstructive airway disease. The treatments include a combination of Budesonide and Formoterol Fumarate Dihydrate in various doses, as well as separate inhalations of each medication, and a placebo. The duration of treatment is likely six months based on the column heading, and there is an "End of Treatment" row with final data.*
Figure 1 - image 03
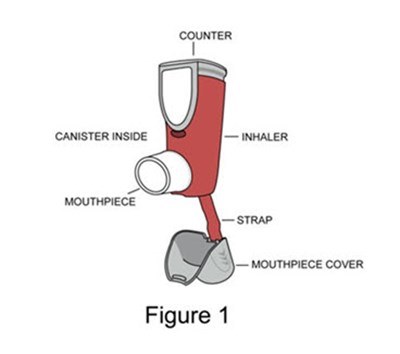
This appears to be a diagram showing the components of a counter with a mouthpiece connected to a strap. The counter seems to have a canister inside with water and a cover for the mouthpiece.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
.jpg)
.jpg)








