Product Images Jardiance
View Photos of Packaging, Labels & Appearance
- Chemical Structure - jardiance 01
- Figure 1 - jardiance 02
- Figure 2 - jardiance 03
- Figure 3 - jardiance 04
- Figure 4 - jardiance 05
- Figure 5 - jardiance 06
- Figure 6 - jardiance 07
- QR code - jardiance 08
- Figure 7 - jardiance 08a
- Figure 8 - jardiance 08b
- Figure 9 - jardiance 08c
- Figure 10 - jardiance 08d
- Figure 11 - jardiance 08e
- Figure 12 - jardiance 08f
- Label Image - lbl500906452
Product Label Images
The following 15 images provide visual information about the product associated with Jardiance NDC 50090-6452 by A-s Medication Solutions, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - jardiance 02
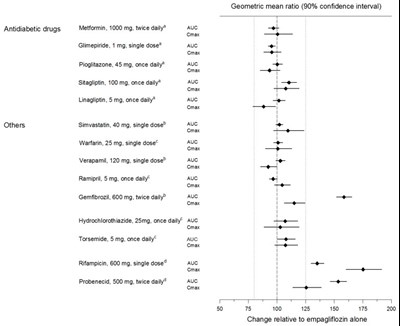
The text refers to a list of antidiabetic drugs with their doses and dosing schedules. It also includes some other drugs such as statins, anticoagulants, blood pressure medications, and others. The text also mentions the geometric mean ratio with a 90% confidence interval. However, there is not enough information to determine the context or purpose of this text.*
Figure 2 - jardiance 03

This is a list of medications with their respective dosages and frequency of administration. The drugs belong to different therapeutic categories such as antidiabetic, oral contraceptives, and others. The list includes Metformin, Glimepiride, Pioglitazone, Sitagliptin, Linagliptin, Etninyestradia, Levonorgestrel, Simvastatin, Warfarin, Ramipril, Ramipriat, Digoxin, Hydrochlorothiazide, and Torsemide. Additionally, the list shows the Cmax (maximum serum concentration) and Auc (area under the curve) values of each medication, as well as the geometric mean ratio (90% confidence interval) for different doses.*
Figure 3 - jardiance 04
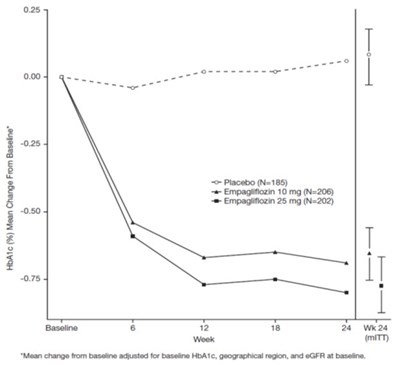
The text describes a chart or figure showing the mean change from baseline HbA1C levels in patients receiving different treatments, including placebo, and two different doses of Empagliflozin, over the course of 24 weeks. The chart also displays the baseline and adjusts the mean change from baseline for certain factors.*
Figure 4 - jardiance 05

The text describes the mean change in HbA1c (%) from baseline for two different drug treatments, Glimepiride + Metformin and Empagliflozin 25 mg + Metformin. The data is based on a sample of 700 individuals for Glimepiride + Metformin and 693 individuals for Empagliflozin 25 mg + Metformin, measured at various time intervals from baseline up to 52 weeks. The mean change is adjusted for baseline HbA1c, geographical region, and eGFR at baseline. Not available.*
Figure 5 - jardiance 06
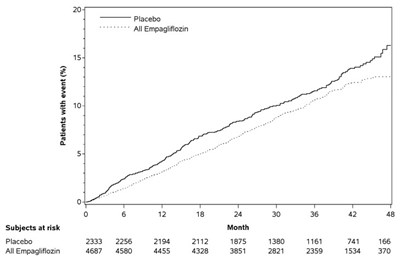
This appears to be a table displaying the percentage of patients who experienced an event, as well as the number of subjects at risk, for a study involving a placebo and a drug called Empaglifiozin. However, the data is not complete and some values are missing, so a full evaluation cannot be made.*
Figure 7 - jardiance 08a
This is the result of a clinical trial comparing Empagliflozin 10mg versus placebo. The table shows the incidence function of % over time, with the placebo group having 1867 participants and the Empagliflozin group also having 1863 participants. The HR (Hazard Ratio) is given for Empagliflozin 10mg versus placebo with statistical significance (P-value) less than 0.0001. The number of patients at risk over time is also provided in each group. This information can be used to evaluate the effectiveness and safety of Empagliflozin.*
Figure 9 - jardiance 08c
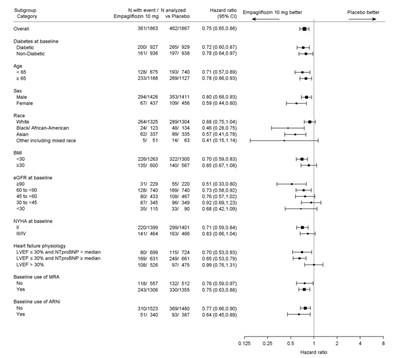
This is a clinical study comparing the effectiveness of Empagliflozin 10mg vs Placebo for diabetes and heart failure patients. The study includes various subgroups defined by diabetes status, sex, race, BMI, GFR, NYHA and heart failure physiology. The Hazard ratios for each group were calculated. Furthermore, the study also evaluated the effect of baseline use of MRA and ARNI on the outcome.*
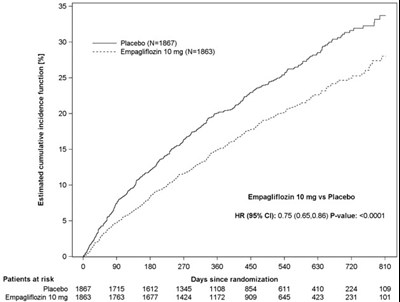
Figure 10 - jardiance 08d
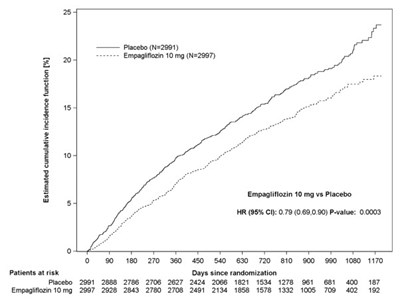
This appears to be a tabular representation of a clinical trial study. It includes values such as incidence function, placebo and Empagiflozin 10 mg. The HR, 95% CI and P value are also mentioned along with a graph showing the number of patients at risk over days since randomization. These details suggest that the text is related to clinical research.*
Figure 11 - jardiance 08e
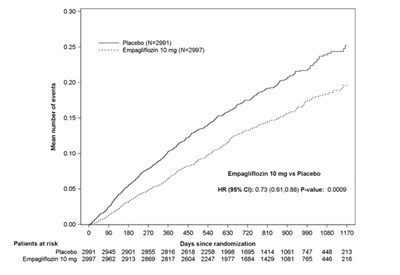
This appears to be a graph or table containing numerical data comparing the effectiveness of a medication called Empagliflozin 10mg (N=2997) versus a placebo (N=2991) for an unknown medical condition. The data appears to show the mean number of events for each treatment group and the number of patients at risk over time since starting the treatment. Additionally, there is a p-value of 0.0009 and a Hazard Ratio of 0.73 (95% confidence interval: 0.61-0.89) comparing Empagliflozin to placebo. However, without further context or information it is difficult to provide a more specific description of what this data represents.*
Figure 12 - jardiance 08f
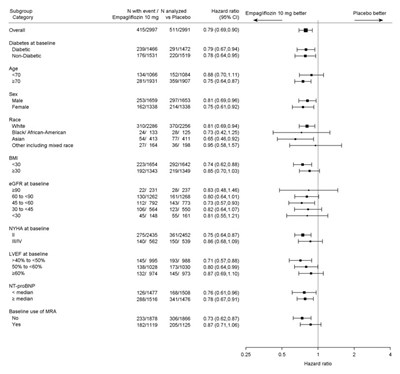
The text appears to be a statistical report on the comparison of the efficacy of Empagliflozin 10mg versus Placebo in reducing events related to diabetes in individuals with and without diabetes. The report provides details on the sample size, diabetes status, sex, race, and baseline conditions of the participants. It also appears to include information on the baseline use of MRA.*
Label Image - lbl500906452

This is a prescription drug with the name "EMPAGLIFLOZIN" and a product code "No. 9939-0". The package contains 90 tablets, each of which contains 16 mg of the active ingredient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



