Product Images Tarina 24 Fe
View Photos of Packaging, Labels & Appearance
- Figure-1 - norethindroneethinyl fig1
- Figure-2 - norethindroneethinyl fig2
- Figure - norethindroneethinyl fig3
- figure4 - norethindroneethinyl fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1 mg/20 mcg Pouch Label - norethindroneethinyl fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1 mg/20 mcg Pouch Carton Label - norethindroneethinyl fig6
- norethindroneethinyl fig7
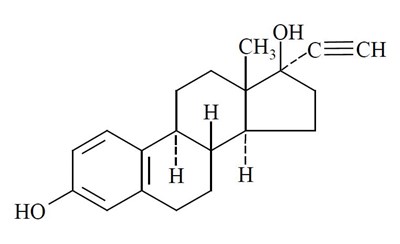
- Structure-1 - norethindroneethinyl str1
- Structure-2 - norethindroneethinyl str2
Product Label Images
The following 9 images provide visual information about the product associated with Tarina 24 Fe NDC 50102-224 by Afaxys Pharma, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure-1 - norethindroneethinyl fig1

This appears to be a list of studies comparing the effects of using combined oral contraceptives (COC) to not using them (never-use) on various health outcomes. The studies listed include the NICHD Women's Care Study, French E3N cohort Study, Shanghai Women's Health Study, The Nurses' Health Study, Oxford Family Planning Study, and Danish Sex Hormone Register Study. The table at the bottom shows effect estimates for the different studies, but without more context, it is difficult to interpret the numbers.*
figure4 - norethindroneethinyl fig4

This text provides information on different birth control methods and their effectiveness in preventing pregnancy. It includes statistics on the number of pregnancies per 100 women in one year for various methods of birth control, including hormonal options like implants and pills, barrier methods like condoms and diaphragms, and non-hormonal methods like spermicide and withdrawal. The text also mentions natural family planning methods like avoiding sex during the most fertile days of the monthly cycle.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1 mg/20 mcg Pouch Carton Label - norethindroneethinyl fig6

Tarinaf® 24 Fe is a pharmaceutical product that provides 24 days of active therapy. It consists of Norethindrone Acetate and Ethinyl Estradiol Tablets USP and Ferrous Fumarate Tablets. Each pouch contains detailed patient labeling, brief summary, patient package insert and day label stickers along with the blister. A 28-day regimen is recommended with one tablet including Combination Detaled Patient Labefing / Brief Summary. Proper storage conditions are mentioned for the product. Overall, Tarinaf® 24 Fe helps with female therapy.*
norethindroneethinyl fig7

Tarina® 24 Fo is an oral contraceptive product consisting of 24 days of active therapy. The product is manufactured by Afaxys Pharma for Aurobindo Pharma Limited in India. The product's NDC is 50102-224-23. It should be noted that Tarina® 24 Fo is not intended to protect against HIV infection or other sexually transmitted diseases. The text includes additional information that is difficult to understand.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.