Product Images Bupropion Hydrochloride SR
View Photos of Packaging, Labels & Appearance
- bupropion HCL 100 mg 1,000 Extended-Release Tablet, USP Label - bupropion 100mg 1000tab
- bupropion HCL 100 mg 100 Extended-Release Tablet, USP Label - bupropion 100mg 100tab
- bupropion HCL 100 mg 30 Extended-Release Tablet, USP Label - bupropion 100mg 30tab
- bupropion HCL 100 mg 500 Extended-Release Tablet, USP Label - bupropion 100mg 500tab
- bupropion HCL 100 mg 60 Extended-Release Tablet, USP Labell - bupropion 100mg 60tab
- bupropion HCL 150 mg 1,000 Extended-Release Tablet, USP Label - bupropion 150mg 1000tab
- bupropion HCL 150 mg 100 Extended-Release Tablet, USP Label - bupropion 150mg 100tab
- bupropion HCL 150 mg 30 Extended-Release Tablet, USP Label - bupropion 150mg 30tab
- bupropion HCL 150 mg 500 Extended-Release Tablet, USP Label - bupropion 150mg 500tab
- bupropion HCL 150 mg 60 Extended-Release Tablet, USP Label - bupropion 150mg 60tab
- bupropion HCL 200 mg 1,000 Extended-Release Tablet, USP Label - bupropion 200mg 1000tab
- bupropion-200mg-100tab - bupropion 200mg 100tab
- bupropion HCL 200 mg 30 Extended-Release Tablet, USP Label - bupropion 200mg 30tab
- bupropion HCL 200 mg 60 Extended-Release Tablet, USP Label - bupropion 200mg 60tab
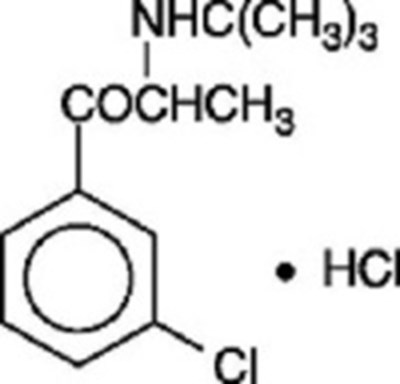
- Chemical Structure - bupropion hydrochloride sr 1
Product Label Images
The following 15 images provide visual information about the product associated with Bupropion Hydrochloride SR NDC 50228-174 by Sciegen Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
bupropion HCL 100 mg 100 Extended-Release Tablet, USP Label - bupropion 100mg 100tab

This text describes a medication called Exoh, which is an extended-release tablet containing 100 mg of bupropion hydrochloride. It is recommended to be taken twice a day after an initial titration. The usual dosage and further details are available in the accompanying prescribing information. The tablets should be stored at room temperature between 68° and 77°F and protected from light and moisture. This medication should be kept out of reach of children and should not be used in combination with ZYBAN or other medicines containing bupropion hydrochloride. Exoh is manufactured by ScieGen Pharmaceuticals and comes in a container with a child-resistant closure.*
bupropion HCL 100 mg 30 Extended-Release Tablet, USP Label - bupropion 100mg 30tab

This text contains information about a medication called "buPRO" which contains bupropion hydrochloride. It is available in extended-release tablets with a dosage of 100mg. Patients should be warned not to use buPRO if they are already taking medications containing bupropion hydrochloride. The medication should be stored at room temperature and away from light and moisture. It is important to keep it out of reach of children. The medication is manufactured by ScieGen Pharmaceuticals in Hauppauge, NY. The expiry date is not available.*
bupropion HCL 100 mg 60 Extended-Release Tablet, USP Labell - bupropion 100mg 60tab

This is a medication information for a drug called "BuPROPion HCI" that comes in an extended-release tablet form containing 100 mg of the drug. It is recommended to consume twice a day, but the dosage should be followed as mentioned in the accompanying prescription. The medication should be kept at room temperature, stored in a tight, light-resistant container with a child-resistant closure to protect it from moisture. The medication guide should be provided to patients with each dispense by pharmacists. Zyban is a registered trademark of GlaxoSmithKline Pharmaceuticals. The medicine should not be used in combination with Zyban or any other medicine containing bupropion hydrochloride. The manufacturer is ScieGen Pharmaceuticals, Inc., located in Hauppauge, NY 11788.*
bupropion HCL 150 mg 1,000 Extended-Release Tablet, USP Label - bupropion 150mg 1000tab

This appears to be a prescription drug bottle label with dosage instructions for using buPROPion HCI extended-release tablets twice a day after initial titration. The label contains a warning not to use the medication in combination with other drugs containing bupropion hydrochloride, and the pharmacist is instructed to dispense an accompanying medication guide to each patient. The label also includes a product code and other identifying information.*
bupropion HCL 150 mg 100 Extended-Release Tablet, USP Label - bupropion 150mg 100tab

This text is a medication label for a drug containing bupropion hydrochloride with advised dosage and storage conditions, along with warnings to keep out of reach of children and not to use with other medications containing bupropion hydrochloride. The drug is manufactured by ScieGen Pharmaceuticals and marketed under the registered trademark ZYBAN.*
bupropion HCL 150 mg 30 Extended-Release Tablet, USP Label - bupropion 150mg 30tab

This is a medication guide for Extended-Release Tablets, USP (SR) containing bupropion hydrochloride, USP, and is to be dispensed by a pharmacist. Patients are warned not to use it with ZYBAN®. The usual dosage and other prescribing information are included. The medication should be stored at controlled room temperature in a light-resistant container with a child-resistant closure. It is manufactured by ScieGen Pharmaceuticals, Inc in the USA.*
bupropion HCL 150 mg 500 Extended-Release Tablet, USP Label - bupropion 150mg 500tab

This is a medication called buPROPion HCI Extended-Release Tablets, USP (SR) used for treating depression and helping people quit smoking. Each tablet has 150 mg of bupropion hydrochloride, USP. It should be stored at 20° to 25°C (66° to 77°F) in a light-resistant container with a child-resistant closure. It's important to keep it away from children at all times. This medication should be taken twice a day after initial medication. It is manufactured by ScieGen Pharmaceuticals, Inc. Pharmacist should dispense the medication guide to each patient. It cannot be used in combination with ZYBAN or other medicines that contain buproprion hydrochloride. Rev. 0420.*
bupropion HCL 150 mg 60 Extended-Release Tablet, USP Label - bupropion 150mg 60tab

This is a medication called "bupropion hydrochloride" that comes in an extended-release tablet container with a strength of 150mg. It is taken twice a day after an initial titration. The usual dosage is not given, and it must be read in the accompanying prescribing information. It should be stored at room temperature, protected from light and moisture, and kept away from children. It comes in a tight container with child-resistant closure. The medication guide must be dispensed to each patient, and it should not be used in combination with ZYBAN or other medicines containing bupropion hydrochloride. ZYBAN is a registered trademark of GlaxoSmitKie. The manufacturer of the medication is ScieGen Pharmaceuticals, located in Hauppauge, NY 11788.*
bupropion-200mg-100tab - bupropion 200mg 100tab

This is a medication called Bupropion Hydrochloride, which is an extended-release tablet that contains 200mg of the active ingredient. The tablet is meant to be taken twice a day and comes with accompanying prescribing information. It should be stored between 20°C-25°C (68°F-77°F) in a light-resistant container with a child-resistant closure. The medication should be protected from light and moisture, and kept out of reach of children. The accompanying medication guide should be dispensed to each patient. The medication should not be used in combination with other medications that contain Bupropion Hydrochloride. The manufacturer is ScieGen Pharmaceuticals, Inc., located in Hauppauge, NY.*
bupropion HCL 200 mg 30 Extended-Release Tablet, USP Label - bupropion 200mg 30tab

This is a medication guide for Ion HCI extended-release tablets USP (SR) containing 200mg bupropion hydrochloride. The tablets should be taken twice a day. Patients should not combine with Zyban or any other medicine that contains bupropion hydrochloride. The recommended dosage should be read in the accompanying prescription information. The tablets should be stored in a tight light-resistant container away from light and moisture at a temperature between 68°F and 77°F. The drug should be kept away from children. The product is manufactured by ScieGen Pharmaceuticals, Inc in Hauppauge, NY 11788.*
bupropion HCL 200 mg 60 Extended-Release Tablet, USP Label - bupropion 200mg 60tab

This is a prescription medication containing 200mg of bupropion hydrochloride, which is to be taken twice daily after an initial titration. It comes in a tight light-resistant container with a child-resistant cap and should be protected from light and moisture. It is important to keep this medication out of reach of children. The medication guide accompanying the product should be dispensed by the pharmacist to each patient. This medication should not be used in combination with ZYBAN or any other medicines containing bupropion hydrochloride. It is manufactured by ScieGen Pharmaceuticals, Inc. and comes in a package of 60 tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.