Product Images Lucentis
View Photos of Packaging, Labels & Appearance
- Figure - lucentis 01
- Figure - lucentis 02
- Figure - lucentis 03
- Figure - lucentis 04
- Figure 1 - lucentis 05
- Figure 2 - lucentis 06
- Figure 3 - lucentis 07
- Figure 4 - lucentis 08
- Figure 5 - lucentis 09
- Figure - lucentis 10
- Figure - lucentis 11
- Figure - lucentis 12
- Figure - lucentis 13
- Figure - lucentis 14
- Figure 1 - lucentis 15
- Figure 2 - lucentis 16
- Figure 3 - lucentis 17
- Figure 4 - lucentis 18
- Figure 5 - lucentis 19
- Figure 6 - lucentis 20
- Figure 7 - lucentis 21
- Figure 8 - lucentis 22
- Figure 9 - lucentis 23
- PRINCIPAL DISPLAY PANEL - 0.5 mg Syringe Label - lucentis 24
- PRINCIPAL DISPLAY PANEL - 0.3 mg Syringe Packet - lucentis 25
Product Label Images
The following 25 images provide visual information about the product associated with Lucentis NDC 50242-080 by Genentech, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - lucentis 05

The text describes a syringe cap with a rubber stopper and finger grip, along with a Luerlock 0.05 mL dose mark plunger rod. The accompanying figure shows what the items look like.*
Figure - lucentis 14

This text refers to a small syringe with a volume capacity of 0.05 milliliters.*
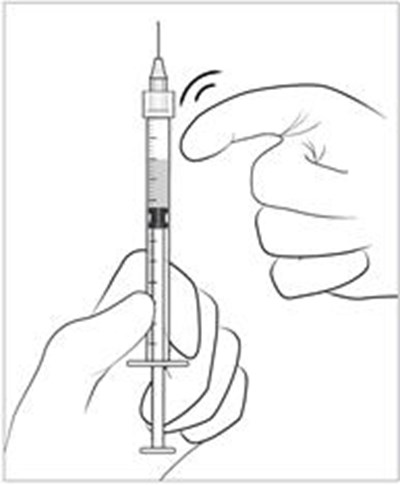
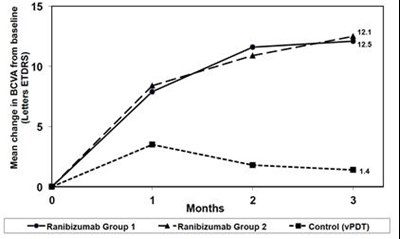
Figure 1 - lucentis 15

Mean Change in Visual Activity for a condition or study named AMD-1 is being measured in meters or getters.*
PRINCIPAL DISPLAY PANEL - 0.5 mg Syringe Label - lucentis 24

This is a description of a drug product called Lucentis. It is a sterile, single-use syringe used for intravitreal injection only. The drug is indicated for the treatment of wet age-related macular degeneration (WAMD), macular edema following retinal vein occlusion (RVO), and myopic choroidal neovascularization (mCNV). The syringe contains 0.5mg of ranibizumab. It is important to keep the product refrigerated and protected from light. It is manufactured by Genentech and is identified by the NDC code 50242.080-03.*
PRINCIPAL DISPLAY PANEL - 0.3 mg Syringe Packet - lucentis 25

This is a description of a medication called Lucentis (ranibizumab injection) that comes in a pre-filled syringe with a 0.3 mg dose. It is used for intravitreal injection only and is indicated for treating diabetic macular edema (DME) and diabetic retinopathy (DR). The medication is sterile, for single use only and should be kept refrigerated, protected from light, and not frozen. The text also includes the National Drug Code (NDC) number and an identification number (10198958).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.