Product Images Tecentriq
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Tecentriq NDC 50242-917 by Genentech, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - tecentriq 02
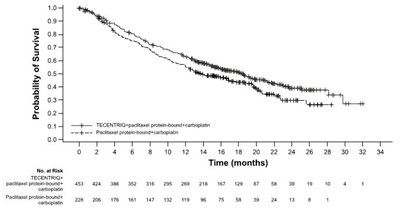
This text seems to describe a graph showing the probability of survival after chemotherapy treatment using TECENTRIO and standard chemotherapy. The x-axis denotes the timeline in months, and the y-axis denotes the probability of survival. There are two lines plotted on the graph. The first line shows the probability of survival for patients treated with TECENTRIO, while the second line shows the probability of survival for patients treated with standard chemotherapy. However, without context or additional information, it is difficult to interpret the graph further.*
Figure 6 - tecentriq 06

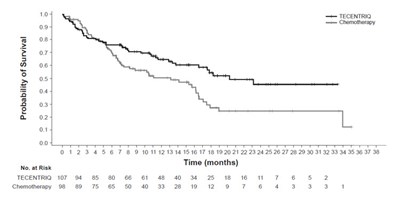
The text describes a chart or graph indicating the Overall Survival percentage over time in months, with comparative treatments shown for TECENTRIQ with Carboplatin and Etoposide, and Placebo with Carboplatin and Etoposide. The horizontal axis shows the Risk Time in months, and the vertical axis shows the Overall Survival percentage. There are multiple values over time for both TECENTRIQ and Placebo, with the highest overall survival.*
Figure 7 - tecentriq 07
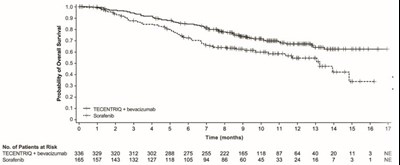
This appears to be a chart displaying data related to the effectiveness of two different treatments on a group of patients over the course of several months. The treatments are listed as "TECENTRIQ+ bovecizumab" and "Sorafond". The chart shows the number of patients who remained under the treatment at each point in time (measured in months) and how many patients dropped out or experienced negative outcomes (indicated by the label "NE").*
Figure 8 - tecentriq 08

The given text is related to two drug combinations TECENTRIQ cobimetit vemurafents and cobmetinbsvemurlend. It also includes a graph displaying the probability of progression-free survival against time (in months). The table shows the number of patients remaining at risk for each of the given treatments. The description of the drugs' properties, usage or efficacy is not available, in the given text.*
PRINCIPAL DISPLAY PANEL - 20 mL Vial Carton - tecentriq 09

This is a product label for the medication Tecentriq® (atezolizumab). It is administered through injection and is meant for intravenous infusion after dilution. It comes in single-dose vials with no preservative. Any unused portion of the medication should be discarded. Pharmacists should dispense the accompanying medication guide to patients. The label also contains the NDC number, which is 50242-917-01, and a code number. The name of the manufacturer, Genentech, is also mentioned. Therefore, this text provides important information about the medication and its administration.*
PRINCIPAL DISPLAY PANEL - 14 mL Vial Carton - tecentriq 10

This is a prescription drug called Tecentriq® with the active ingredient atezolizumab. It is administered through an injection for intravenous infusion after dilution and comes in a single-dose vial. The unused portion should be discarded, and there is no preservative. The accompanying medication guide must be dispensed to every patient by the pharmacist. The drug is manufactured by Genentech and has the NDC code 50242-918-01.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.