Product Images Hemlibra
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 47 images provide visual information about the product associated with Hemlibra NDC 50242-922 by Genentech, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - hemlibra 01

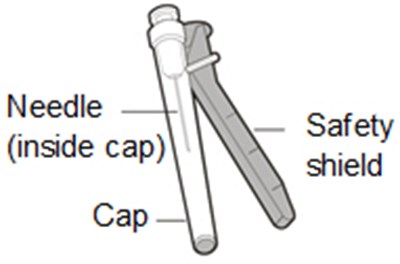
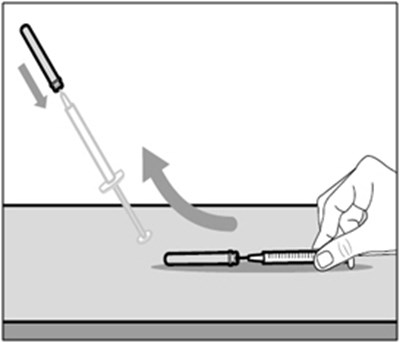
This text describes a Transfer Needle with Filter that is used specifically for transferring the medication HEMLIBRA from a vial to a syringe. The product consists of a needle and cap with an enclosed filter.*
Figure - hemlibra 06

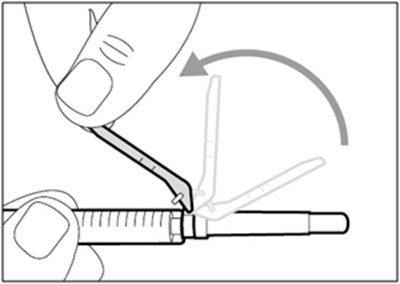
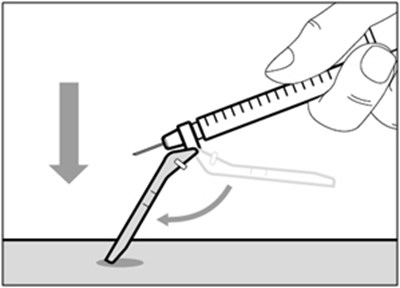
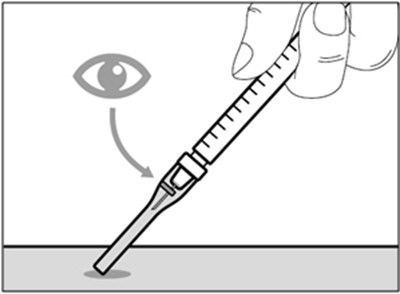
This is not enough information to generate a useful description. The text seems to be labels for parts or products, but there is no context or further information available.*
Figure - hemlibra 10

Upper arm is a body part located between the shoulder and elbow joints. It is made up of bone, muscle, and connective tissue, and is responsible for providing strength and mobility to the shoulder and elbow joints.*
PRINCIPAL DISPLAY PANEL - 30 mg Vial Carton - hemlibra 42

Hemlibra® is a medication that can be injected subcutaneously using a single-dose vial. Each vial contains 30 mg/mL of emicizumab-kxwh. It is important to discard any unused portions after administration. The medication guide enclosed should be dispensed to each patient. The NDC number for the medication is 50242-920-01 and it is manufactured by Genentech.*
PRINCIPAL DISPLAY PANEL - 60 mg Vial Carton - hemlibra 43

Hemlibra is a medication for subcutaneous use that comes in a single-dose vial containing 60mg/0.4mL of emicizumab-kxwh. The product should be discarded if any remaining quantity is leftover. The enclosed medication guide should be given to each patient.*
PRINCIPAL DISPLAY PANEL - 105 mg Vial Carton - hemlibra 44

Hemlibra is a medication available in a single-dose vial for subcutaneous injection. It contains emicizumab-kxwh and is used to treat hemophilia. A medication guide should be included and unused portions should be discarded.*
PRINCIPAL DISPLAY PANEL - 150 mg Vial Carton - hemlibra 45

Hemlibra is a medication used through subcutaneous injection. It comes in single-dose vials containing 150 mg/mL of emicizumab-kxwh. The medication guide enclosed with the product must be dispensed to each patient. This text appears to be a label for the medication, including the NDC number and product name.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.