Product Images Clopidogrel
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Clopidogrel NDC 50268-184 by Avpak, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
300mg - 184 12

Clopidogrel Tablets, USP are a prescription medication containing 391.5 mg of clopidogrel bisulfate, USP equivalent to 300 mg of clopidogrel. They come in 20 tablets (4 x 5) unit dose and are meant to be dispensed by a pharmacist with an accompanying medication guide to each patient. These tablets are manufactured by AVKARE, Inc. and should be kept at a controlled room temperature within the range of 15°C to 30°C (59°F to 86°F). The usual adult dosage should be determined by the physician and described in the prescribing information.*
Figure 3 - clopidogrel 04

This is a chart showing the hazard ratio for patient baseline characteristics and on-study concomitant medications in the CURE study. The chart includes subgroups based on certain criteria such as gender, race, diabetes, previous medical history, and concomitant medications. There are also hazard ratios listed for each subgroup.*
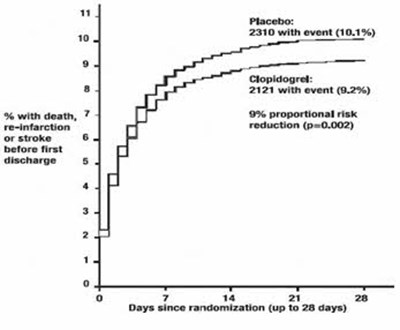
Figure 4 - clopidogrel 05

This text appears to be a chart or graph which shows the percentage of deaths before first discharge for a group taking a placebo. The chart includes the number of deaths (1845) and the percentage of deaths (8.1%). The x-axis shows the number of days since randomization, up to 28 days. Additional information is not available.*
Figure 6 - clopidogrel 07

This is a statistical table presenting the results of a study on the use of Clopidogrel compared to a placebo. The study assessed the efficacy of Clopidogrel in different subgroups of patients classified by gender, age, hours since onset, systolic blood pressure, heart rate, and fibrinolytic agent given. The table displays the number of patients in each subgroup as well as the percentage of participants who received either Clopidogrel or the placebo. The last row shows the overall results of the study and the corresponding odds ratio.*
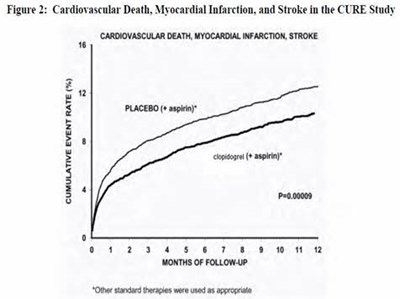
Figure 7 - clopidogrel 08

This is a graph showing the cumulative event rate (in percentage) of fatal or non-fatal vascular events between aspirin and clopidogrel over 18 months of follow-up. The y-axis ranges from 0 to 16% and the x-axis shows the months of follow-up. The data suggests that clopidogrel is associated with a lower cumulative event rate compared to aspirin. The statistical significance of the difference is indicated with p=0.045 at the bottom of the graph.*
Figure 8 - clopidogrel 09

This is a statistical table showing the results of a study comparing the efficacy of Clopidogrel and Aspirin in treating stroke patients. The table provides data on the number and percentage of patients who received each medication, as well as the hazard ratios for each year of the study. The hazard ratio represents the likelihood of an event occurring in one group compared to another. However, given the limited information, it is impossible to provide a more detailed description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.