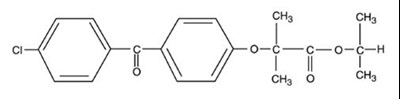
Product Images Fenofibrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Fenofibrate NDC 50268-338 by Avpak, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
20 - 338 12

This is a description of medication with NDC 50260.338-12 identification number. The drug is Fenofibrate and is available in a pack of 20 tablets with two unit dosages. This medicine contains 4mg of Exchtabltconin enfbrta USP. Dosage varies and must be given according to the accompanying prescription. It should be stored at 25° (77), with excursions permitted by 1510 S0 (50 t085°F [0 USP Controlod Room Tompartrl Potet fom moltre. The medication should be kept out of reach of children, and its details indicate that it is only accessible by prescription. The medication is manufactured for AVKARE, and the packaging comes from P, TH 35478 AVRRK MG Rox.*
4 - Table 4

The table displays the mean percent change in lipid parameters at the end of treatment. The table has three treatment groups categorized by baseline lipid values with different Total-C, LDLC, and HDL-C parameters. The percent change in lipid parameters, including Total-C, LDLC, and HDL-C, are displayed for each group, along with their baseline lipid values in mg/dL. The study treatment had a significant effect compared to the placebo.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.