Product Images Methylphenidate Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Methylphenidate Hydrochloride NDC 50268-546 by Avpak, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
18 - 545 12 18mg

This is a product label and medication guide for Methylphenidate Hydrochloride Extended-Release Tablets, a medication available only by prescription. The tablets are sold in unit dose packaging, with each containing 18mg of methylphenidate hydrochloride in a controlled-release formulation. The usual dosage is once a day, and the pharmacist is advised to provide patients with an enclosed medication guide. The tablets should be stored at controlled room temperature and away from humidity. The medication is manufactured by Avkare in Pulaski, TN.*
27 - 546 12 27mg

This is a description of Methylphenidate Hydrochloride C Extended-Release Tablets, USP, with a usual dosage of once daily. Each tablet contains 27mg of methylphenidate hydrochloride in a controlled-release formulation. The package comes with 20 tablets in a unit dose of 4 x 5, available for dispensing through prescription only. The pharmacist must dispense the enclosed Medication Guide to each patient, and the tablets should be stored at 20° to 25°C (68° to 77°). Methylphenidate is a type of medication used to treat attention deficit hyperactivity disorder (ADHD) and narcolepsy.*
36 - 547 12 36mg

Methylphenidate Hydrochloride C Extended-Release Tablets, USP 36 mg is a prescription medication used to treat attention deficit hyperactivity disorder. Each tablet contains 36 mg of methylphenidate hydrochloride in a controlled-release formulation, and the usual dosage is once a day. The medication comes in a pack of 20 tablets (4 x 5) and should be stored between 20°C to 25°C (68°F to 77°F), protected from humidity. A medication guide is enclosed to be dispensed to each patient, and the product should be kept out of reach of children. Manufactured for Avkare, Pulaski, TN 38478.*
54 - 548 12 54mg

This is a description of a medication called Methylphenidate hydrochloride, which comes in 54 mg Extended-Release Tablets. The medication is only available by prescription, and there are 20 tablets in each package. The medication guide enclosed should be given to each patient by the pharmacist and the tablets should be stored between 20° to 25°C (68° to 77°F), protected from humidity, and kept away from children. It's manufactured for AVKARE in Pulaski, TN 38478. See package insert for dosage information.*
TB1 - methylphenidate table1

This text provides recommended starting doses and dose ranges for methylphenidate hydrochloride extended-release tablets based on the patient's age. It includes recommended starting doses and dose ranges for children, adolescents, and adults. In particular, the recommended starting dose for children aged 6-12 is 18 mg/day, while adolescents aged 13-17 are also recommended 18 mg/day, not to exceed 2 mg/kg/day. For adults aged 18-65, the recommended starting dose is 18 or 36 mg/day, with a dose range of 18 mg - 72 mg/day. This information may be useful for healthcare professionals prescribing medications to patients for attention deficit hyperactivity disorder (ADHD).*
Table3 - methylphenidate table3

This is a table that shows the exposure of Methylphenidate hydrochloride extended-release tablets in double-blind and open-label clinical studies. It provides information on the patient population, number of patients studied, and the dose range administered to each population. It shows that the medication was administered to 2216 children, 502 adolescents, and 1188 adults at varying doses once daily.*
Table 4 - methylphenidate table4

Table 4 provides a breakdown of adverse reactions reported in four placebo-controlled, double-blind clinical trials of Methylphenidate Hydrochloride Extended Release tablets among children and adolescents. The table lists the percentages of subjects who reported specific adverse reactions, such as abdominal pain, dizziness, cough, and insomnia, among others. These reactions were reported by more than 1% of the treated subjects.*
Tb 6 - methylphenidate table6

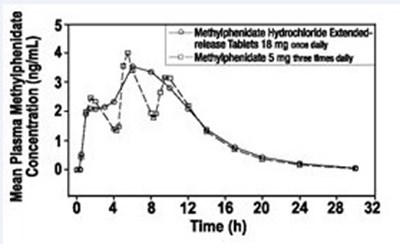
Table 6 shows the pharmacokinetic parameters such as Cou (ng/mL), Tinex, AUCq¢ (ngeh/mL), and ta after a single dose of Methylphenidate hydrochloride extended-release tablets in healthy adults. The parameters for a dose of 5 mg three times daily and 18 mg once daily are also included.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.