Product Images Buprenorphine And Naloxone
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Buprenorphine And Naloxone NDC 50383-294 by Akorn Operating Company Llc (dba Akorn), such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Bottle Label - 0.5mg/2mg - buprenorphine naloxone 04

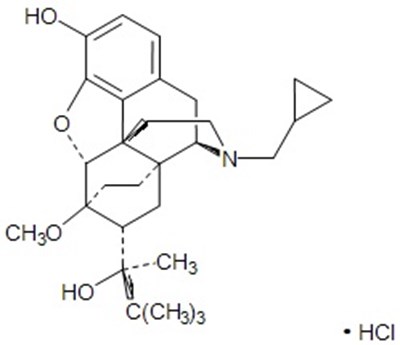
Each tablet contains Buprenorphine 216mg and Naloxone 0.61mg as a sublingual Tablet used as per Product Information for medical purposes, accompanied by a Medication Guide for pharmacist use only. Produced by Ethypharm S.A in France and distributed in the US by Akom Operating Company LLC. The tablets should be stored at 20° to 25°C (68° to 77°F) in a controlled room temperature setting, and the cap should be kept out of reach of children to avoid accidental ingestion. If children consume the drug, immediate medical care is necessary. No other useful information is available in the given text.*
Bottle Label - 8mg/2mg - buprenorphine naloxone 05

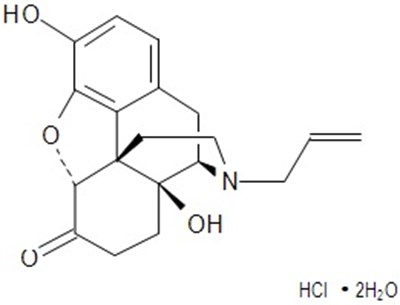
This is a description of a medication which is a combination of buprenorphine HCI and naloxone HCI dihydrate. The tablets are manufactured by Ethypharm S.A. in France and distributed by Akorn Operating Company LLC in Gurnee, Illinois. Each tablet contains 8.64mg of buprenorphine and 2.44mg of naloxone. The recommended dosage and storage information is provided in the insert included with the product. The medication is dispensed with a medication guide which includes important safety information. The medication is prescription-only and requires medical care in the event of accidental ingestion by children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.