Product Images Adempas
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Adempas NDC 50419-254 by Bayer Healthcare Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Effect of Intrinsic Factors on Riociguat and M1 Pharmacokinetics - e9e1a668 1b31 4094 88b7 2004f94af53d 02

Figure 2: Effect of Extrinsic Factors on Riociguat and M1 Pharmacokinetics - e9e1a668 1b31 4094 88b7 2004f94af53d 03

This text appears to be a list of medications and substances, along with recommendations for potential interactions with other drugs or supplements. The list includes various strong and moderate inhibitors and inducers, as well as antacids, antiplatelet agents, and more. The specific details of the recommendations are difficult to decipher without further context, but it appears that some medications may require dose adjustments or separate administration to avoid potential negative interactions.*
Figure 5: Mean Treatment Difference in Change from Baseline to Last Visit in 6-Minute Walk Distance (meters) by Prespecified Subgroups - e9e1a668 1b31 4094 88b7 2004f94af53d 06

PATENT-1 Mean Change from Baseline in the 6-Minute Walk Distance - e9e1a668 1b31 4094 88b7 2004f94af53d 07

PATENT-1 Distribution of Patients by Change from Baseline in 6-Minute Walk Distan - e9e1a668 1b31 4094 88b7 2004f94af53d 08

PATENT-1 Mean Treatment Difference in Change from Baseline to Last Visit in 6-Minute Walk Distance (meter) by Prespecified Subgroups - e9e1a668 1b31 4094 88b7 2004f94af53d 09

PATENT-1 Time (in Days) to Clinical Worsening (ITT analysis set) - e9e1a668 1b31 4094 88b7 2004f94af53d 10

.5 carton - e9e1a668 1b31 4094 88b7 2004f94af53d 12

Adempas is a medication that comes in tablet form, containing Riociguat. The dosage should be strictly followed, and it needs to be stored at 25 degree Celsius. It is a prescription drug, and the details about the drug, such as composition and storage, are provided on the carton. The provided text contains some distortion; might have not read it correctly.*
2 Carton - e9e1a668 1b31 4094 88b7 2004f94af53d 15

This is a description of a medication called Adempas, which comes in tablet form. The tablets are available in a strength of 2mg per tablet and are embossed with the code "PR S2277060". The medication contains riociguat and is used to treat pulmonary arterial hypertension. The label on the packaging mentions that the tablets are varnish-free, but there is no information available on what this means.*
2.5 carton - e9e1a668 1b31 4094 88b7 2004f94af53d 16

This text contains information on a medicine called Adempas (riociguat) tablets. Each container has 30 soluble tablets, containing 25 mg of riociguat per tablet. The recommended dosage is to take one tablet three times daily. Storage instructions advise keeping the medicine at room temperature (25-77 F) and protecting it from light. The text also includes various product codes and references to a "VARNISH FREE" feature, but it is unclear how this relates to the medicine itself.*
Figure 4: CHEST-1 Distribution of Patients by Change from Baseline in 6-Minute Walk Distance - image 01
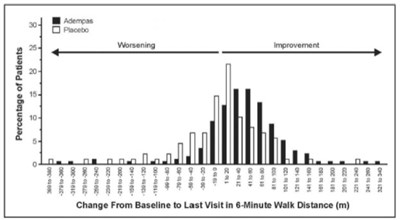
This seems to be a table or chart depicting the change in 6-minute walk distance in some sort of study or experiment. The text also includes the words "Improvement" and "Worsening" which likely indicate the direction of change in the participants' walking ability. However, the rest of the text appears to be a mix of letters, numbers, and symbols that cannot be interpreted as readable language.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




