FDA Label for Skyla
View Indications, Usage & Precautions
- 1 INDICATIONS AND USAGE
- 2 DOSAGE AND ADMINISTRATION
- 2.1. INSERTION INSTRUCTIONS
- TIMING OF INSERTION
- PREPARATION
- PROCEDURE
- PREPARATION FOR INSERTION
- INSERTION PROCEDURE
- STEP 1–OPENING OF THE PACKAGE
- STEP 2–LOAD SKYLA INTO THE INSERTION TUBE
- STEP 3–SETTING THE FLANGE
- STEP 4–SKYLA IS NOW READY TO BE INSERTED
- STEP 5–OPEN THE ARMS
- STEP 6–ADVANCE TO FUNDAL POSITION
- STEP 7–RELEASE SKYLA AND WITHDRAW THE INSERTER
- IMPORTANT INFORMATION TO CONSIDER DURING OR AFTER INSERTION
- 2.2 PATIENT FOLLOW-UP
- TIMING OF REMOVAL
- REMOVAL PROCEDURE
- 2.4 CONTINUATION OF CONTRACEPTION AFTER REMOVAL
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5.1 RISKS OF ECTOPIC PREGNANCY
- 5.2 RISKS WITH INTRAUTERINE PREGNANCY
- SEPTIC ABORTION
- CONTINUATION OF PREGNANCY
- 5.3 SEPSIS
- PELVIC INFLAMMATORY DISEASE (PID)
- WOMEN AT INCREASED RISK FOR PID
- SUBCLINICAL PID
- TREATMENT OF PID
- ACTINOMYCOSIS
- 5.5 PERFORATION
- 5.6 EXPULSION
- 5.7 OVARIAN CYSTS
- 5.8 BLEEDING PATTERN ALTERATIONS
- 5.9 BREAST CANCER
- 5.10 CLINICAL CONSIDERATIONS FOR USE AND REMOVAL
- 5.11 MAGNETIC RESONANCE IMAGING (MRI) SAFETY INFORMATION
- 6 ADVERSE REACTIONS
- 6.1 CLINICAL TRIALS EXPERIENCE
- 6.2 POSTMARKETING EXPERIENCE
- 7 DRUG INTERACTIONS
- RISK SUMMARY
- 8.4 PEDIATRIC USE
- 8.5 GERIATRIC USE
- 11 DESCRIPTION
- 11.1 SKYLA
- 11.2 INSERTER
- 12.1 MECHANISM OF ACTION
- 12.2 PHARMACODYNAMICS
- ABSORPTION
- DISTRIBUTION
- ELIMINATION
- METABOLISM
- EXCRETION
- SPECIFIC POPULATIONS
- DRUG-DRUG INTERACTIONS
- 13.1 CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY
- 14 CLINICAL STUDIES
- 15 REFERENCES
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
- SPL PATIENT PACKAGE INSERT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Skyla Product Label
The following document was submitted to the FDA by the labeler of this product Bayer Healthcare Pharmaceuticals Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
1 Indications And Usage
Skyla® is indicated to prevent pregnancy for up to 3 years.
Replace the system after 3 years if continued use is desired.
2 Dosage And Administration
Skyla contains 13.5 mg of levonorgestrel (LNG) released in vivo at a rate of approximately 14 mcg/day after 24 days. This rate decreases progressively to approximately 6 mcg/day after 1 year and to 5 mcg/day after 3 years. The average in vivo release rate of LNG is approximately 8 mcg/day over the first year of use and 6 mcg/day over a period of 3 years. [See Clinical Pharmacology (12.3).]
Skyla must be removed by the end of the third year and can be replaced at the time of removal with a new Skyla if continued contraceptive protection is desired.
Skyla can be physically distinguished from other intrauterine systems (IUSs) by the combination of the visibility of the silver ring on ultrasound and the brown color of the removal threads.
Skyla is supplied in a sterile package within an inserter that enables single-handed loading (see Figure 1). Do not open the package until required for insertion [see Description (11)]. Do not use if the seal of the sterile package is broken or appears compromised. Use strict aseptic techniques throughout the insertion procedure [see Warnings and Precautions (5.3)].
2.1. Insertion Instructions
• Obtain a complete medical and social history to determine conditions that might influence the selection of a levonorgestrel-releasing intrauterine system (LNG IUS) for contraception. If indicated, perform a physical examination, and appropriate tests for any forms of genital or other sexually transmitted infections. [See Contraindications (4) and Warnings and Precautions (5.10).]• Follow the insertion instructions exactly as described to ensure proper placement and avoid premature release of Skyla from the inserter. Once released, Skyla cannot be re-loaded.• Skyla should be inserted by a trained healthcare provider. Healthcare providers should become thoroughly familiar with the insertion instructions before attempting insertion of Skyla.• Insertion may be associated with some pain and/or bleeding or vasovagal reactions (for example, syncope, bradycardia) or with seizure, especially in patients with a predisposition to these conditions. Consider administering analgesics prior to insertion.
Timing Of Insertion
Table 1: When to Insert Skyla
Starting Skyla in women not currently using hormonal or intrauterine contraception |
|
Switching to Skyla from an oral, transdermal or vaginal hormonal contraceptive |
|
Switching to Skyla from an injectable progestin contraceptive |
|
Switching to Skyla from a contraceptive implant or another IUS |
|
Inserting Skyla after abortion or miscarriage | |
First-trimester |
|
Second-trimester |
|
Inserting Skyla after Childbirth |
|
Preparation
• Gloves• Speculum• Sterile uterine sound• Sterile tenaculum• Antiseptic solution, applicator• Gloves• Speculum
Procedure
• Sterile gloves• Skyla with inserter in sealed package• Instruments and anesthesia for paracervical block, if anticipated• Consider having an unopened backup Skyla available• Sterile, sharp curved scissors• Sterile forceps
Preparation For Insertion
• Exclude pregnancy and confirm that there are no other contraindications to the use of Skyla.• Ensure that the patient understands the contents of the Patient Information Booklet and obtain the signed patient informed consent located on the last page of the Patient Information Booklet.• Check expiration date of Skyla prior to initiating insertion.• With the patient comfortably in lithotomy position, do a bimanual exam to establish the size, shape and position of the uterus.• Gently insert a speculum to visualize the cervix.• Thoroughly cleanse the cervix and vagina with a suitable antiseptic solution.• Prepare to sound the uterine cavity. Grasp the upper lip of the cervix with a tenaculum forceps and gently apply traction to stabilize and align the cervical canal with the uterine cavity. Perform a paracervical block if needed. If the uterus is retroverted, it may be more appropriate to grasp the lower lip of the cervix. The tenaculum should remain in position and gentle traction on the cervix should be maintained throughout the insertion procedure.• Gently insert a uterine sound to check the patency of the cervix, measure the depth of the uterine cavity in centimeters, confirm cavity direction, and detect the presence of any uterine anomaly. If you encounter difficulty or cervical stenosis, use dilatation, and not force, to overcome resistance. If cervical dilatation is required, consider using a paracervical block.
Insertion Procedure
Proceed with insertion only after completing the above steps and ascertaining that the patient is appropriate for Skyla. Ensure use of aseptic technique throughout the entire procedure.
Step 1–Opening Of The Package
• Open the package (Figure 1). The contents of the package are sterile.• Using sterile gloves, lift the handle of the sterile inserter and remove from the sterile package.
Step 2–Load Skyla Into The Insertion Tube
• Push the slider forward as far as possible in the direction of the arrow thereby moving the insertion tube over the Skyla T-body to load Skyla into the insertion tube (Figure 2). The tips of the arms will meet to form a rounded end that extends slightly beyond the insertion tube.• Maintain forward pressure with your thumb or forefinger on the slider. DO NOT move the slider downward at this time as this may prematurely release the threads of Skyla. Once the slider is moved below the mark, Skyla cannot be re-loaded.
Step 3–Setting The Flange
• Holding the slider in this forward position, set the upper edge of the flange to correspond to the uterine depth (in centimeters) measured during sounding (Figure 3).
Step 4–Skyla Is Now Ready To Be Inserted
• Continue holding the slider in this forward position. Advance the inserter through the cervix until the flange is approximately 1.5–2 cm from the cervix and then pause (Figure 4).
Do not force the inserter. If necessary, dilate the cervical canal.
Step 5–Open The Arms
• While holding the inserter steady, move the slider down to the mark to release the arms of Skyla (Figure 5). Wait 10 seconds for the horizontal arms to open completely.
Step 6–Advance To Fundal Position
Advance the inserter gently towards the fundus of the uterus until the flange touches the cervix. If you encounter fundal resistance do not continue to advance. Skyla is now in the fundal position (Figure 6). Fundal positioning of Skyla is important to prevent expulsion.
Step 7–Release Skyla And Withdraw The Inserter
• Holding the entire inserter firmly in place, release Skyla by moving the slider all the way down (Figure 7).• Continue to hold the slider all the way down while you slowly and gently withdraw the inserter from the uterus.• Using a sharp, curved scissor, cut the threads perpendicular, leaving about 3 cm visible outside of the cervix [cutting threads at an angle may leave sharp ends (Figure 8)]. Do not apply tension or pull on the threads when cutting to prevent displacing Skyla.
.
Important Information To Consider During Or After Insertion
• If you suspect that Skyla is not in the correct position, check placement (for example, using transvaginal ultrasound). Remove Skyla if it is not positioned completely within the uterus. Do not reinsert a removed Skyla.• If there is clinical concern, exceptional pain or bleeding during or after insertion, take appropriate steps (such as physical examination and ultrasound) immediately to exclude perforation.
2.2 Patient Follow-Up
• Reexamine and evaluate patients 4 to 6 weeks after insertion and once a year thereafter, or more frequently if clinically indicated.
Timing Of Removal
• Skyla should not remain in the uterus after 3 years.• If pregnancy is not desired, the removal should be carried out during the first 7 days of the menstrual cycle, provided the woman is experiencing regular menses. If removal will occur at other times during the cycle or the woman does not experience regular menses, she is at risk of pregnancy: start a new contraceptive method a week prior to removal for these women. [See Dosage and Administration (2.4).]
Removal Procedure
• Remove Skyla by applying gentle traction on the threads with forceps (Figure 9).• If the threads are not visible, determine location of Skyla by ultrasound [see Warnings and Precautions (5.10)].• If Skyla is found to be in the uterine cavity on ultrasound exam, it may be removed using a narrow forceps, such as an alligator forceps. This may require dilation of the cervical canal. After removal of Skyla, examine the system to ensure that it is intact.• Removal may be associated with some pain and/or bleeding or vasovagal reactions (for example, syncope, bradycardia) or seizure, especially in patients with a predisposition to these conditions.
2.4 Continuation Of Contraception After Removal
• If pregnancy is not desired and if a woman wishes to continue using Skyla, a new system can be inserted immediately after removal any time during the cycle.• If a patient with regular cycles wants to start a different contraceptive method, time removal and initiation of the new method to ensure continuous contraception. Either remove Skyla during the first 7 days of the menstrual cycle and start the new method immediately thereafter or start the new method at least 7 days prior to removing Skyla if removal is to occur at other times during the cycle.• If a patient with irregular cycles or amenorrhea wants to start a different contraceptive method, start the new method at least 7 days before removal.
3 Dosage Forms And Strengths
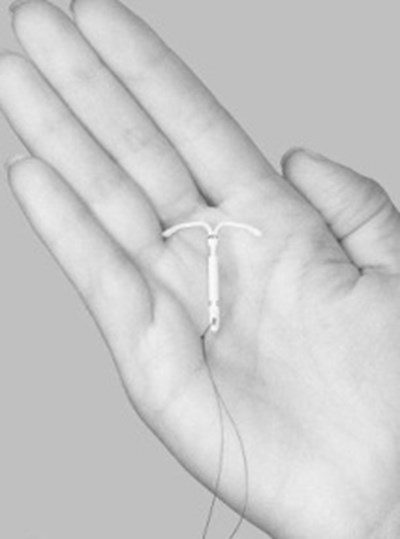
Skyla is a LNG-releasing IUS (a type of intrauterine device, or IUD) consisting of a T-shaped polyethylene frame with a steroid reservoir containing a total of 13.5 mg LNG.
4 Contraindications
The use of Skyla is contraindicated when one or more of the following conditions exist:
• Pregnancy or suspicion of pregnancy [see Warnings and Precautions (5.2), Use in Specific Populations (8.1)]• For use as post-coital contraception (emergency contraception)• Congenital or acquired uterine anomaly including fibroids, that distorts the uterine cavity• Acute pelvic inflammatory disease (PID) or a history of PID unless there has been a subsequent intrauterine pregnancy [see Warnings and Precautions (5.4)]• Postpartum endometritis or infected abortion in the past 3 months• Known or suspected uterine or cervical neoplasia• Known or suspected breast cancer or other progestin-sensitive cancer, now or in the past• Uterine bleeding of unknown etiology• Untreated acute cervicitis or vaginitis, including bacterial vaginosis or other lower genital tract infections until infection is controlled• Acute liver disease or liver tumor (benign or malignant)• Conditions associated with increased susceptibility to pelvic infections [see Warnings and Precautions (5.4)]• A previously inserted intrauterine device (IUD) that has not been removed• Hypersensitivity to any component of this product [see Adverse Reactions (6.2) and Description (11.1)]
5.1 Risks Of Ectopic Pregnancy
The incidence of ectopic pregnancy in clinical trials with Skyla, which excluded women with a history of ectopic pregnancy, was approximately 0.1% per year. The risk of ectopic pregnancy in women who have a history of ectopic pregnancy and use Skyla is unknown. Women with a previous history of ectopic pregnancy, tubal surgery or pelvic infection carry a higher risk of ectopic pregnancy. Ectopic pregnancy may result in loss of fertility.
5.2 Risks With Intrauterine Pregnancy
If pregnancy occurs while using Skyla, remove Skyla because leaving it in place may increase the risk of spontaneous abortion and preterm labor. Removal of Skyla or probing of the uterus may also result in spontaneous abortion. In the event of an intrauterine pregnancy with Skyla, consider the following:
Septic Abortion
In patients becoming pregnant with an IUS in place, septic abortion—with septicemia, septic shock, and death—may occur.
Continuation Of Pregnancy
If a woman becomes pregnant with Skyla in place and if Skyla cannot be removed or the woman chooses not to have it removed, warn her that failure to remove Skyla increases the risk of miscarriage, sepsis, premature labor and premature delivery. Follow her pregnancy closely and advise her to report immediately any symptom that suggests complications of the pregnancy.
5.3 Sepsis
Severe infection or sepsis, including Group A streptococcal sepsis (GAS), have been reported following insertion of a LNG-releasing IUS. In some cases, severe pain occurred within hours of insertion followed by sepsis within days. Because death from GAS is more likely if treatment is delayed, it is important to be aware of these rare but serious infections. Aseptic technique during insertion of Skyla is essential in order to minimize serious infections such as GAS.
Pelvic Inflammatory Disease (Pid)
Skyla is contraindicated in the presence of known or suspected PID or in women with a history of PID unless there has been a subsequent intrauterine pregnancy [see Contraindications (4)]. IUDs have been associated with an increased risk of PID, most likely due to organisms being introduced into the uterus during insertion. In clinical trials, PID was observed in 0.4% of women overall and occurred more frequently within the first year and most often within the first month after insertion of Skyla.
Promptly examine users with complaints of lower abdominal or pelvic pain, odorous discharge, unexplained bleeding, fever, genital lesions or sores. Remove Skyla in cases of recurrent endometritis or pelvic inflammatory disease, or if an acute pelvic infection is severe or does not respond to treatment.
Women At Increased Risk For Pid
PID is often associated with a sexually transmitted infection (STI), and Skyla does not protect against STI. The risk of PID is greater for women who have multiple sexual partners, and also for women whose sexual partner(s) have multiple sexual partners. Women who have had PID are at increased risk for a recurrence or re-infection. In particular, ascertain whether the woman is at increased risk of infection (for example, leukemia, acquired immune deficiency syndrome [AIDS], intravenous drug abuse).
Subclinical Pid
PID may be asymptomatic but still result in tubal damage and its sequelae.
Treatment Of Pid
Following a diagnosis of PID, or suspected PID, bacteriologic specimens should be obtained and antibiotic therapy should be initiated promptly. Removal of Skyla after initiation of antibiotic therapy is usually appropriate.
Actinomycosis
Actinomycosis has been associated with IUDs. Remove Skyla from symptomatic women and treat with antibiotics. The significance of actinomyces-like organisms on Pap smear in an asymptomatic IUD user is unknown, and so this finding alone does not always require Skyla removal and treatment. When possible, confirm a Pap smear diagnosis with cultures.
5.5 Perforation
Perforation (total or partial, including penetration/embedment of Skyla in the uterine wall or cervix) may occur most often during insertion, although the perforation may not be detected until sometime later. Perforation may reduce contraceptive efficacy and result in pregnancy. The incidence of perforation during clinical trials was < 0.1%.
If perforation occurs, locate and remove Skyla. Surgery may be required. Delayed detection or removal of Skyla in case of perforation may result in migration outside the uterine cavity, adhesions, peritonitis, intestinal perforations, intestinal obstruction, abscesses and erosion of adjacent viscera.
The risk of perforation may be increased if Skyla is inserted when the uterus is fixed retroverted or not completely involuted. Delay Skyla insertion a minimum of six weeks or until involution is complete following a delivery or a second trimester abortion.
Clinical trials with Skyla excluded breast-feeding women. A large postmarketing safety study conducted in Europe over a 1-year observational period reported that lactation at the time of insertion of an IUD/IUS was associated with an increased risk of perforation. For users of another LNG-releasing IUS, the incidence of uterine perforation was reported as 6.3 per 1,000 insertions for lactating women, compared to 1.0 per 1,000 insertions for non-lactating women.
5.6 Expulsion
Partial or complete expulsion of Skyla may occur resulting in the loss of contraceptive protection. Expulsion may be associated with symptoms of bleeding or pain, or it may be asymptomatic and go unnoticed. Skyla typically decreases menstrual bleeding over time; therefore, an increase of menstrual bleeding may be indicative of an expulsion. The risk of expulsion may be increased when the uterus is not completely involuted. In clinical trials, a 3-year expulsion rate of 3.2% (54 out of 1,665 subjects) was reported.
Delay Skyla insertion a minimum of six weeks or until uterine involution is complete following a delivery or a second trimester abortion. Remove a partially expelled Skyla. If expulsion has occurred, a new Skyla can be inserted any time the provider can be reasonably certain the woman is not pregnant.
5.7 Ovarian Cysts
Because the contraceptive effect of Skyla is mainly due to its local effects within the uterus, ovulatory cycles with follicular rupture usually occur in women of fertile age using Skyla. Ovarian cysts (reported as adverse reactions if they were abnormal, non-functional cysts and/or had a diameter >3 cm on ultrasound examination) were reported at least once over the course of clinical trials in 13.2% of women using Skyla, and 0.3% of subjects discontinued because of an ovarian cyst. Most ovarian cysts are asymptomatic, although some may be accompanied by pelvic pain or dyspareunia. In most cases the ovarian cysts disappear spontaneously during two to three months observation. Evaluate persistent ovarian cysts. Surgical intervention is not usually required.
5.8 Bleeding Pattern Alterations
Skyla can alter the bleeding pattern and result in spotting, irregular bleeding, heavy bleeding, oligomenorrhea and amenorrhea. During the first 3–6 months of Skyla use, the number of bleeding and spotting days may be higher and bleeding patterns may be irregular. Thereafter, the number of bleeding and spotting days usually decreases but bleeding may remain irregular.
In Skyla clinical trials, amenorrhea developed by the end of the first year of use in approximately 6% of Skyla users. A total of 77 subjects out of 1,672 (4.6%) discontinued due to uterine bleeding complaints. Table 2 shows the bleeding patterns as documented in the Skyla clinical trials based on 90-day reference periods. Table 3 shows the number of bleeding and spotting days based on 28-day cycle equivalents.
Skyla | First 90 days | Second 90 days | End of year 1 | End of year 3 |
Amenorrhea1 | <1% | 3% | 6% | 12% |
Infrequent bleeding2 | 8% | 19% | 20% | 22% |
Frequent bleeding3 | 31% | 12% | 8% | 4% |
Prolonged bleeding4 | 55% | 14% | 6% | 2% |
Irregular bleeding5 | 39% | 25% | 18% | 15% |
1Defined as subjects with no bleeding/spotting throughout the 90-day reference period
2Defined as subjects with 1 or 2 bleeding/spotting episodes in the 90-day reference period
3Defined as subjects with more than 5 bleeding/spotting episodes in the 90-day reference period
4Defined as subjects with bleeding/spotting episodes lasting more than 14 days in the 90-day reference period. Subjects with prolonged bleeding may also be included in one of the other categories (excluding amenorrhea)
5Defined as subjects with 3 to 5 bleeding/spotting episodes and less than 3 bleeding/spotting-free intervals of 14 or more days.
28-day Cycle Equivalent | Cycle 1 N=1,588 | Cycle 4 N=1,535 | Cycle 7 N=1,468 | Cycle 13 N=1,345 | Cycle 39 N=781 |
Days on treatment | 1–28 | 85–112 | 169–196 | 337–364 | 1065–1092 |
Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | |
Number of bleeding days | 7.3 (5.6) | 3.5 (3.4) | 2.8 (3.1) | 2.1 (2.7) | 1.4 (2.1) |
Number of spotting days | 9.2 (6.1) | 4.8 (4.4) | 3.8 (3.6) | 3.3 (3.1) | 2.7 (2.7) |
Because irregular bleeding/spotting is common during the first months of Skyla use, exclude endometrial pathology (polyps or cancer) prior to the insertion of Skyla in women with persistent or uncharacteristic bleeding. If a significant change in bleeding develops during prolonged use, take appropriate diagnostic measures to rule out endometrial pathology. Consider the possibility of pregnancy if menstruation does not occur within six weeks of the onset of a previous menstruation. Once pregnancy has been excluded, repeated pregnancy tests are generally not necessary in amenorrheic women unless indicated, for example, by other signs of pregnancy or by pelvic pain.
5.9 Breast Cancer
Women who currently have or have had breast cancer, or have a suspicion of breast cancer, should not use hormonal contraception, including Skyla because some breast cancers are hormone-sensitive [see Contraindications (4)].
Spontaneous reports of breast cancer have been received during postmarketing experience with a LNG-releasing IUS. Observational studies of the risk of breast cancer with use of a LNG-releasing IUS do not provide conclusive evidence of increased risk.
5.10 Clinical Considerations For Use And Removal
Use Skyla with caution after careful assessment if any of the following conditions exist, and consider removal of the system if any of them arise during use:
• Coagulopathy or use of anticoagulants• Migraine, focal migraine with asymmetrical visual loss or other symptoms indicating transient cerebral ischemia• Exceptionally severe headache• Marked increase of blood pressure• Severe arterial disease such as stroke or myocardial infarction• Uterine or cervical malignancy• Jaundice• If the threads are not visible or are significantly shortened they may have broken or retracted into the cervical canal or uterus. Consider the possibility that the system may have been displaced, (for example, expelled or perforated the uterus) [see Warnings and Precautions (5.5, 5.6)]. Exclude pregnancy and verify the location of Skyla, for example, by sonography, X-ray, or by gentle exploration of the cervical canal with a suitable instrument. If Skyla is displaced, remove it. A new Skyla may be inserted at that time or during the next menses if it is certain that conception has not occurred. If Skyla is in place with no evidence of perforation, no intervention is indicated.
In addition, consider removing Skyla if any of the following conditions arise during use:
5.11 Magnetic Resonance Imaging (Mri) Safety Information
Non-clinical testing has demonstrated that Skyla is MR Conditional. A patient with Skyla can be safely scanned in an MR system meeting the following conditions:
• Static magnetic field of 3.0 T or less• Maximum spatial field gradient of 36,000 gauss/cm (360 T/m)• Maximum MR system reported, whole body averaged specific absorption rate (SAR) of 4W/kg (First Level Controlled Operating Mode)
Under the scan conditions defined above, the Skyla IUS is expected to produce a maximum temperature rise of less than 2°C after 15 minutes of continuous scanning.
In non-clinical testing, the image artifact caused by the IUS extended up to 5 mm from the IUS when imaged with a gradient echo pulse sequence and a 3.0 T MRI system.
6 Adverse Reactions
The following serious or otherwise important adverse reactions are discussed elsewhere in the labeling:
• Ectopic Pregnancy [see Warnings and Precautions (5.1)]• Intrauterine Pregnancy [see Warnings and Precautions (5.2)]• Group A Streptococcal Sepsis (GAS) [see Warnings and Precautions (5.3)]• Pelvic Inflammatory Disease [see Warnings and Precautions (5.4)]• Perforation [see Warnings and Precautions (5.5)]• Expulsion [see Warnings and Precautions (5.6)]• Ovarian Cysts [see Warnings and Precautions (5.7)]• Bleeding Pattern Alterations [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The data described below reflect exposure to Skyla in 1,672 patients in two contraception studies, including 1,383 exposed for one year and 993 who completed the three year studies. The population was generally healthy, 18 to 40-year old females requesting contraception and predominately Caucasian (82.6%). The data cover more than 40,000 cycles of exposure. The frequencies of reported adverse drug reactions represent crude incidences.
Most common adverse reactions (occurring in ≥ 5% users) were increased bleeding (7.8%), vulvovaginitis (20.2%), abdominal/pelvic pain (18.9%), acne/seborrhea (15.0%), ovarian cyst (13.2%), headache (12.4%), dysmenorrhea (8.6%), breast pain/discomfort (8.6%) and nausea (5.5%).
In the contraception studies, 18% discontinued prematurely due to an adverse reaction. The most common adverse reactions leading to discontinuation (in >1% of users) were uterine bleeding complaints (4.6%), device expulsion (3.2%), acne/seborrhea (2.9%), abdominal pain (2.5%) dysmenorrhea/uterine spasms (2.0%) and pelvic pain (1.8%).
Other common adverse reactions (occurring in ≥ 1% users) by System Organ Class (SOC): The frequencies of adverse reactions observed in clinical trials are summarized in Table 3 by SOC (presented as crude incidences).
System Organ Class | Adverse Reaction | Incidence (%) |
Reproductive System and Breast Disorders |
|
|
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
|
| |
Gastrointestinal Disorders |
|
|
|
| |
Skin and Subcutaneous |
|
|
|
| |
Nervous System Disorders |
|
|
|
| |
Psychiatric Disorders |
|
|
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of LNG-releasing IUSs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
• Arterial thrombotic and venous thromboembolic events, including cases of pulmonary embolism, deep vein thrombosis and stroke• Device breakage• Hypersensitivity (including rash, urticaria, and angioedema)• Increased blood pressure
7 Drug Interactions
No drug-drug interaction studies have been conducted with Skyla.
Drugs or herbal products that induce or inhibit LNG metabolizing enzymes, including CYP3A4, may decrease or increase, respectively, the serum concentrations of LNG during the use of Skyla. However, the contraceptive effect of Skyla is mediated via the direct release of LNG into the uterine cavity and is unlikely to be affected by drug interactions via enzyme induction or inhibition.
Risk Summary
The use of Skyla is contraindicated in pregnancy or with a suspected pregnancy because there is no need for pregnancy prevention in a woman who is already pregnant and Skyla may cause adverse pregnancy outcomes [see Contraindications (4), Warnings and Precautions (5.1, 5.2)]. If a woman becomes pregnant with Skyla in place, the likelihood of ectopic pregnancy is increased and there is an increased risk of miscarriage, sepsis, premature labor, and premature delivery. Remove Skyla, if possible, if pregnancy occurs in a woman using Skyla. If Skyla cannot be removed, follow the pregnancy closely [see Warnings and Precautions (5.1, 5.2)].
Studies report no adverse effects on fetal and infant development associated with long-term use of contraceptive doses of oral progestins in a pregnant woman. However, there have been reported cases of masculinization of the external genitalia of the female fetus following exposure to progestins at doses greater than those currently used for oral contraception. Animal reproduction studies have not been conducted with Skyla.
Published studies report the presence of LNG in human milk. Small amounts of progestins (approximately 0.1% of the total maternal doses) were detected in the breast milk of nursing mothers who used other LNG-releasing IUSs, resulting in exposure of LNG to the breastfed infants. There are no reports of adverse effects in breastfed infants with maternal use of progestin-only contraceptives. Isolated cases of decreased milk production have been reported with a LNG-releasing IUS. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Skyla and any potential adverse effects on the breastfed child from Skyla or from the underlying maternal condition.
8.4 Pediatric Use
Safety and efficacy of Skyla have been established in women of reproductive age. Efficacy is expected to be the same for postpubertal females under the age of 18 as for users 18 years and older. Use of this product before menarche is not indicated.
8.5 Geriatric Use
Skyla has not been studied in women over age 65 and is not approved for use in this population.
11 Description
Skyla (levonorgestrel-releasing intrauterine system) contains 13.5 mg of LNG, a progestin, and is intended to provide an initial release rate of approximately14 mcg/day of LNG after 24 days.
Levonorgestrel USP, (-)-13-Ethyl-17-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one, the active ingredient in Skyla, has a molecular weight of 312.4, a molecular formula of C21H28O2, and the following structural formula:
11.1 Skyla
Skyla consists of a T-shaped polyethylene frame (T-body) with a steroid reservoir (hormone elastomer core) around the vertical stem. The white T-body has a loop at one end of the vertical stem and two horizontal arms at the other end. The reservoir consists of a whitish or pale yellow cylinder, made of a mixture of LNG and silicone (polydimethylsiloxane), containing a total of 13.5 mg LNG. The reservoir is covered by a semi-opaque silicone membrane, composed of polydimethylsiloxane and colloidal silica. A ring composed of 99.95% pure silver is located at the top of the vertical stem close to the horizontal arms and is visible by ultrasound. The polyethylene of the T-body is compounded with barium sulfate, which makes it radiopaque. A monofilament brown polyethylene removal thread is attached to a loop at the end of the vertical stem of the T-body. The polyethylene of the removal thread contains iron oxide as a colorant (see Figure 10).
The components of Skyla, including its packaging, are not manufactured using natural rubber latex.
11.2 Inserter
Skyla is packaged sterile within an inserter. The inserter (Figure 11), which is used for insertion of Skyla into the uterine cavity, consists of a symmetric two-sided body and slider that are integrated with flange, lock, pre-bent insertion tube and plunger. The outer diameter of the insertion tube is 3.8 mm. The vertical stem of Skyla is loaded in the insertion tube at the tip of the inserter. The arms are pre-aligned in the horizontal position. The removal threads are contained within the insertion tube and handle. Once Skyla has been placed, the inserter is discarded.
12.1 Mechanism Of Action
The local mechanism by which continuously released LNG contributes to the contraceptive effectiveness of Skyla has not been conclusively demonstrated. Studies of Skyla and similar LNG IUS prototypes have suggested several mechanisms that prevent pregnancy: thickening of cervical mucus preventing passage of sperm into the uterus, inhibition of sperm capacitation or survival, and alteration of the endometrium.
12.2 Pharmacodynamics
Skyla has mainly local progestogenic effects in the uterine cavity. The local concentrations of LNG lead to morphological changes including stromal pseudodecidualization, glandular atrophy, a leukocytic infiltration and a decrease in glandular and stromal mitoses.
In clinical trials with Skyla, ovulation was assessed based on serum progesterone values >2.5 ng/mL in one study and serum progesterone values >2.5 ng/mL together with serum estradiol levels <27.24 pg/mL in another study. Evidence of ovulation by these criteria was seen in 34 out of 35 women in the first year, in 26 out of 27 women in the second year, and in all 26 women in the third year.
Absorption
Low doses of LNG are administered into the uterine cavity with the Skyla intrauterine delivery system. The in vivo release rate is approximately 14 mcg/day after 24 days and is reduced to approximately 10 mcg/day after 60 days and then declines progressively to approximately 6 mcg/day after 1 year and 5 mcg/day after 3 years. The average LNG in vivo release rate is approximately 8 mcg/day over the first year of use and 6 mcg/day over the period of 3 years.
In a subset of 7 subjects, the maximum observed serum LNG concentration (mean ±SD) was 192 ± 105 pg/mL, reached after 2 days (median) of Skyla insertion. Thereafter, the LNG serum concentrations (mean ±SD) at year 1, 2, and 3 were 77 ± 21 pg/mL, 62 ± 38 pg/mL, and 72 ± 29 pg/mL, respectively. A population pharmacokinetic evaluation based on a broader database (>1000 patients) showed similar concentration data of 168 ± 46 pg/mL at 7 days after placement. Thereafter, LNG serum concentrations decline slowly to a value 61 ± 19 pg/mL after 3 years.
Distribution
The apparent volume of distribution of LNG is reported to be approximately 1.8 L/kg. LNG is bound non-specifically to serum albumin and specifically to sex hormone binding globulin (SHBG). Accordingly, changes in the concentration of SHBG in serum result in an increase (at higher SHBG concentration) or a decrease (at lower SHBG concentration) of the total LNG concentration in serum. In a subset of 7 subjects, the concentration of SHBG declined by a mean value of 18% within 2 weeks after insertion of Skyla and remained relatively stable over the 3 year period of use. Less than 2% of the circulating LNG is present as free steroid.
Elimination
Following intravenous administration of 0.09 mg LNG to healthy volunteers, the total clearance of LNG is approximately 1 mL/min/kg and the elimination half-life is approximately 20 hours. Metabolic clearance rates may differ among individuals by several-fold, and this may account in part for wide individual variations in LNG concentrations seen in individuals using LNG–containing contraceptive products.
Metabolism
Excretion
LNG and its phase I metabolites are excreted primarily as glucuronide conjugates. About 45% of LNG and its metabolites are excreted in the urine and about 32% are excreted in feces, mostly as glucuronide conjugates.
Specific Populations
Pediatric: Safety and efficacy of Skyla have been established in women of reproductive age. Use of this product before menarche is not indicated.
In a one-year phase 3 study in post-menarcheal female adolescents (mean age 16.2, range 12 to 18 years) using Skyla, the population pharmacokinetic analysis of 278 adolescents showed mean estimated LNG serum concentrations slightly higher (approximately 10%) in adolescents compared to prior data in adults. This correlates to the generally lower body weight in adolescents. The ranges estimated for adolescents lie within the ranges estimated for adults.
Geriatric: Skyla has not been studied in women over age 65 and is not approved for use in this population.
Race: A three-year phase 3 study in the Asian-Pacific region (93% Asian women, the majority of whom were Chinese, 7% other ethnicities) using Skyla was performed. The population pharmacokinetic analysis of the Asian (Chinese) population in this study showed that estimated LNG serum concentrations in Asian women were slightly higher (approximately 4 to 16%) than those in another phase 3 study which was performed in mainly Caucasian women (79.7%). This slightly higher exposure might be explained by the lower body weight of Asian women.
Hepatic Impairment: No studies were conducted to evaluate the effect of hepatic disease on the disposition of Skyla.
Renal Impairment: No formal studies were conducted to evaluate the effect of renal disease on the disposition of Skyla.
Drug-Drug Interactions
No drug-drug interaction studies were conducted with Skyla [see Drug Interactions (7)].
13.1 Carcinogenesis, Mutagenesis, Impairment Of Fertility
[See Warnings and Precautions (5.9).]
14 Clinical Studies
The contraceptive efficacy of Skyla was evaluated in a clinical trial that enrolled generally healthy women aged 18–35, 1,432 of whom received Skyla. Of these, 38.8 % (556) were nulliparous women, and 819 women completed 3 years of use. The trial was a multicenter, multi-national, randomized open-label study conducted in 11 countries in Europe, Latin America, the US and Canada. Women less than six weeks postpartum, with a history of ectopic pregnancy, with clinically significant ovarian cysts or with HIV or otherwise at high risk for sexually transmitted infections were excluded. A total of 540 (37.7%) were treated at US sites and 892 (62.3%) were at non-US sites. The racial demographics of enrolled women who received Skyla was: Caucasian (79.7%), Hispanic (11.5%), Black/African Americans (5.2%), Other (2.7%) and Asian (0.8%). The weight range was 38 to 155 kg (mean weight: 68.7 kg) and mean BMI was 25.3 kg/m2 (range 16–55 kg/m2). The clinical trial had no upper or lower weight or BMI limit. Of Skyla-treated women, 21.9% discontinued the study treatment due to an adverse event, 4.4% were lost to follow up, 1.8% withdrew for unspecified reasons, 1.1% discontinued due to protocol deviation, 0.6% discontinued due to pregnancy, and 13.0% discontinued due to other reasons.
The pregnancy rate calculated as the Pearl Index (PI) in women aged 18–35 years was the primary efficacy endpoint used to assess contraceptive reliability. The PI was calculated based on 28-day equivalent exposure cycles; evaluable cycles excluded those in which back-up contraception was used unless a pregnancy occurred in that cycle. Skyla-treated women provided 15,763 evaluable 28-day cycle equivalents in the first year and 39,368 evaluable cycles over the three year treatment period. The PI estimate for the first year of use based on the 5 pregnancies that occurred after the onset of treatment and within 7 days after Skyla removal or expulsion was 0.41 with a 95% upper confidence limit of 0.96. The cumulative 3-year pregnancy rate, based on 10 pregnancies, estimated by the Kaplan-Meier method was 0.9 per 100 women or 0.9%, with a 95% upper confidence limit of 1.7%.
About 77% of women who desired pregnancy after study discontinuation and provided follow-up information, conceived within 12 months after removal of Skyla.
15 References
1http://www.cdc.gov/std/tg2015/pid.htm. Accessed August 22, 2016.
16 How Supplied/Storage And Handling
Skyla is supplied sterile. Skyla is sterilized with ethylene oxide. Do not resterilize. For single use only. Do not use if the inner package is damaged or open. Insert before the end of the month shown on the label.
Store at 25°C (77°F); with excursions permitted between 15–30°C (59–86°F) [see USP Controlled Room Temperature].
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information)
• Sexually Transmitted Infections: Counsel the patient that this product does not protect against HIV infection (AIDS) and other sexually transmitted infections (STIs).• Risk of Ectopic Pregnancy: Inform the patient about the risks of ectopic pregnancy, including the loss of fertility. Teach her to recognize and report to her healthcare provider promptly any symptoms of ectopic pregnancy. [See Warnings and Precautions (5.1).]• Risks of Intrauterine Pregnancy: Instruct the patient to contact her healthcare provider if she thinks she might be pregnant. Inform the patient about the risks of intrauterine pregnancy while using Skyla, including the risks of leaving Skyla in place and the risks of removing Skyla or probing of the uterus. If Skyla cannot be removed in a pregnant patient, advise her to report immediately any symptom that suggests complications of the pregnancy. [See Warnings and Precautions (5.2) and Use in Special Populations (8.1).]• Sepsis: Counsel the patient that severe infection or sepsis, including Group A streptococcal sepsis (GAS), can occur within the first few days after Skyla is inserted. Instruct her to contact a healthcare provider immediately if she develops severe pain or fever shortly after Skyla is inserted. [See Warnings and Precautions (5.3).]• Pelvic Infection: Inform the patient about the possibility of pelvic infections including PID and that these infections can cause tubal damage leading to ectopic pregnancy or infertility, or infrequently can necessitate hysterectomy, or cause death. Teach patients to recognize and report to their healthcare provider promptly any symptoms of pelvic infection. These symptoms include development of menstrual disorders (prolonged or heavy bleeding), unusual vaginal discharge, abdominal or pelvic pain or tenderness, dyspareunia, chills, and fever. [See Warnings and Precautions (5.4).]• Perforation and Expulsion: Counsel the patient on how she can check that the threads still protrude from the cervix and caution her not to pull on the threads and displace Skyla. Inform her that there is no contraceptive protection if Skyla is displaced (for example, expelled or perforated the uterus). If perforation occurs, Skyla will have to be located and removed; surgery may be required. Instruct the patient to contact her healthcare provider if she cannot feel the threads. [See Warnings and Precautions (5.5, 5.6, 5.10).]• Ovarian Cysts: Counsel the patient regarding the risk of ovarian cysts and that cysts can cause clinical symptoms including pelvic pain, abdominal pain or dyspareunia. Advise the patient to contact her healthcare provider if she experiences these symptoms. [See Warnings and Precautions (5.7).]• Bleeding Pattern Alterations: Counsel the patient that irregular or prolonged bleeding and spotting, and/or cramps may occur during the first few weeks after insertion. Inform the patient that, during the first 3–6 months of Skyla use, the number of bleeding and spotting days may be higher and bleeding patterns may be irregular. If her symptoms continue or are severe she should report them to her healthcare provider. [See Warnings and Precautions (5.8).]• Clinical Considerations for Use and Removal: Instruct the patient to contact her healthcare provider if she experiences any of the following:• A stroke or heart attack• Very severe or migraine headaches• Unexplained fever• Yellowing of the skin or whites of the eyes, as these may be signs of serious liver problems• Pregnancy or suspected pregnancy• Pelvic pain, abdominal pain, or pain during sex• HIV positive seroconversion in herself or her partner• Possible exposure to STIs• Unusual vaginal discharge or genital sores• Severe vaginal bleeding or bleeding that lasts a long time, or if she misses a menstrual period• Inability to feel Skyla's threads• Magnetic Resonance Imaging (MRI) Safety Information: Inform the patient that Skyla can be safely scanned with MRI only under specific conditions. Instruct patients who will have an MRI to tell their doctor that they have Skyla. This information is included on the Follow-Up Reminder Card. [See Warnings and Precautions (5.11).]
Complete the Follow-up Reminder Card and give it to the patient.
Spl Patient Package Insert
FDA-Approved Patient Labeling
Patient Information Skyla (sky-lah) (levonorgestrel-releasing intrauterine system) | |
Skyla does not protect against HIV infection (AIDS) and other sexually transmitted infections (STIs). Read this Patient Information carefully before you decide if Skyla is right for you. This information does not take the place of talking with your gynecologist or other healthcare provider who specializes in women’s health. If you have any questions about Skyla, ask your healthcare provider. You should also learn about other birth control methods to choose the one that is best for you. | |
What is Skyla?
Skyla is a small, flexible plastic T-shaped system that slowly releases a progestin hormone called levonorgestrel (LNG) that is often used in birth control pills. Because Skyla releases LNG into your uterus, only small amounts of the hormone enter your blood. Skyla does not contain estrogen. Two thin threads are attached to the stem (lower end) of Skyla. The threads are the only part of Skyla you can feel when Skyla is in your uterus; however, unlike a tampon string, the threads do not extend outside your body. | |
Skyla is small | and flexible |
What if I need birth control for more than 3 years? Skyla must be removed after 3 years. Your healthcare provider can place a new Skyla during the same office visit if you choose to continue using Skyla. What if I want to stop using Skyla? Skyla is intended for use up to 3 years but you can stop using Skyla at any time by asking your healthcare provider to remove it. You could become pregnant as soon as Skyla is removed, so you should use another method of birth control if you do not want to become pregnant. Talk to your healthcare provider about the best birth control methods for you, because your new method may need to be started 7 days before Skyla is removed to prevent pregnancy. What if I change my mind about birth control and want to become pregnant in less than 3 years? Your healthcare provider can remove Skyla at any time. You may become pregnant as soon as Skyla is removed. About 3 out of 4 women who want to become pregnant will become pregnant sometime in the first year after Skyla is removed. | |
How does Skyla work? Skyla may work in several ways including thickening cervical mucus, inhibiting sperm movement, reducing sperm survival, and thinning the lining of your uterus. It is not known exactly how these actions work together to prevent pregnancy. | |
How well does Skyla work for contraception? The following chart shows the chance of getting pregnant for women who use different methods of birth control. Each box on the chart contains a list of birth control methods that are similar in effectiveness. The most effective methods are at the top of the chart. The box on the bottom of the chart shows the chance of getting pregnant for women who do not use birth control and are trying to get pregnant. Skyla, an intrauterine device (IUD), is in the box at the top of the chart. Who might use Skyla? You might choose Skyla if you:
| |
Do not use Skyla if you:
Before having Skyla placed, tell your healthcare provider if you: | |
How is Skyla placed? Skyla is placed by your healthcare provider during an in-office visit. First, your healthcare provider will examine your pelvis to find the exact position of your uterus. Your healthcare provider will then clean your vagina and cervix with an antiseptic solution and slide a slim plastic tube containing Skyla into your uterus. Your healthcare provider will then remove the plastic tube, and leave Skyla in your uterus. Your healthcare provider will cut the threads to the right length. Placement takes only a few minutes. You may experience pain, bleeding or dizziness during and after placement. If your symptoms do not pass within 30 minutes after placement, Skyla may not have been placed correctly. Your healthcare provider will examine you to see if Skyla needs to be removed or replaced. Should I check that Skyla is in place? Yes, you should check that Skyla is in proper position by feeling the removal threads. It is a good habit to do this 1 time a month. Your healthcare provider should teach you how to check that Skyla is in place. First, wash your hands with soap and water. You can check by reaching up to the top of your vagina with clean fingers to feel the removal threads. Do not pull on the threads. If you feel more than just the threads or if you cannot feel the threads, Skyla may not be in the right position and may not prevent pregnancy. Use non-hormonal back-up birth control (such as condoms and spermicide) and ask your healthcare provider to check that Skyla is still in the right place. How soon after placement of Skyla should I return to my healthcare provider? Call your healthcare provider if you have any questions or concerns (see “When should I call my healthcare provider?”). Otherwise, you should return to your healthcare provider for a follow-up visit 4 to 6 weeks after Skyla is placed to make sure that Skyla is in the right position. Can I use tampons with Skyla? Yes tampons may be used with Skyla. What if I become pregnant while using Skyla? Call your healthcare provider right away if you think you may be pregnant. If possible, also do a urine pregnancy test. If you get pregnant while using Skyla, you may have an ectopic pregnancy. This means that the pregnancy is not in the uterus. Unusual vaginal bleeding or abdominal pain may be a sign of ectopic pregnancy. Ectopic pregnancy is a medical emergency that often requires surgery. Ectopic pregnancy can cause internal bleeding, infertility, and even death. There are also risks if you get pregnant while using Skyla and the pregnancy is in the uterus. Severe infection, miscarriage, premature delivery, and even death can occur with pregnancies that continue with an intrauterine device (IUD). Because of this, your healthcare provider may try to remove Skyla, even though removing it may cause a miscarriage. If Skyla cannot be removed, talk with your healthcare provider about the benefits and risks of continuing the pregnancy. If you continue your pregnancy, see your healthcare provider regularly. Call your healthcare provider right away if you get flu-like symptoms, fever, chills, cramping, pain, bleeding, vaginal discharge, or fluid leaking from your vagina. These may be signs of infection. It is not known if Skyla can cause long-term effects on the fetus if it stays in place during a pregnancy. How will Skyla change my periods? For the first 3 to 6 months, your period may become irregular and the number of bleeding days may increase. You may also have frequent spotting or light bleeding. Some women have heavy bleeding during this time. After you have used Skyla for a while, the number of bleeding and spotting days is likely to lessen. For some women, periods will stop altogether. When Skyla is removed, your menstrual periods should return. Is it safe to breastfeed while using Skyla? You may use Skyla when you are breastfeeding if more than 6 weeks have passed since you had your baby. If you are breastfeeding, Skyla is not likely to affect the quality or amount of your breast milk or the health of your nursing baby. However, isolated cases of decreased milk production have been reported. The risk of Skyla becoming attached to (embedded) or going through the wall of the uterus is increased if Skyla is inserted while you are breastfeeding. Will Skyla interfere with sexual intercourse? You and your partner should not feel Skyla during intercourse. Skyla is placed in the uterus, not in the vagina. Sometimes your partner may feel the threads. If this occurs, or if you or your partner experience pain during sex, talk with your healthcare provider. Can I have an MRI with Skyla in place? Skyla can be safely scanned with MRI only under specific conditions. Before you have an MRI, tell your healthcare provider that you have Skyla, an intrauterine device (IUD), in place. | |
What are the possible side effects of Skyla? Skyla can cause serious side effects, including:
Tell your healthcare provider right away if you have any of these signs of PID: long-lasting or heavy bleeding, unusual vaginal discharge, low abdominal (stomach area) pain, painful sex, chills, or fever. | |
Common side effects of Skyla include:
This is not a complete list of possible side effects with Skyla. For more information, ask your healthcare provider. Tell your healthcare provider if you have any side effect that bothers you or does not go away. Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to the manufacturer at 1-888-842-2937 or www.fda.gov/medwatch. After Skyla has been placed, when should I call my healthcare provider? If Skyla is accidentally removed and you had vaginal intercourse within the preceding week, you may be at risk of pregnancy, and you should talk to a healthcare provider. Call your healthcare provider if you have any concerns about Skyla. Be sure to call if you: | |
General advice about the safe and effective use of Skyla Medicines are sometimes prescribed for conditions that are not mentioned in patient information leaflets. You can ask your healthcare provider for information about Skyla that is written for healthcare providers. For more information, go to www.skyla-us.com or call 1-888-842-2937. | |
Manufactured for: Bayer HealthCare Pharmaceuticals Inc., Whippany, NJ 07981 © 2000, Bayer HealthCare Pharmaceuticals Inc. All rights reserved. | |
Revised 3/2018
Package Label.Principal Display Panel
Skyla (levonorgestrel-releasing intrauterine system) Carton
NDC 50419-422-01
1 Sterile Unit
IMPORTANT: To be inserted in the uterus by or under the supervision of a licensed clinician. See physician insert for detailed instructions for use.
Skyla
(levonorgestrel-releasing intrauterine system)
Rx only
— 13.5 mg levonorgestrel
— 1 sterile unit
— intrauterine use
* Please review the disclaimer below.