Topiramate
Product Images NDC 50436-0139
View Photos of Packaging, Regulatory Labels, and Product Appearance
Product Visual Gallery
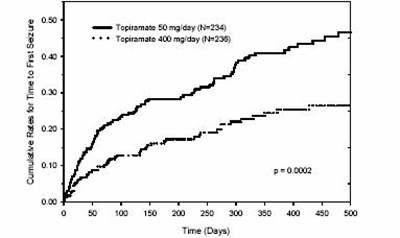
This gallery contains 4 technical images submitted to the FDA as part of the official labeling for Topiramate (NDC 50436-0139). Unlike standard consumer photos, these assets often include clinical data figures, molecular chemical structures, and official manufacturer packaging layouts.
As provided by Unit Dose Services, these visuals offer a comprehensive scientific overview of the product's physical and chemical identity, aiding pharmacists and researchers in product verification and study.
Structured Product Formula For Topiramate (16e9304c A029 4d23 Ae3f E197def9a302 02)
Topiramate Tablets Usp (16e9304c A029 4d23 Ae3f E197def9a302 03)

The text shows the change in the average monthly migraine period rate from baseline to the double-blind phase for different groups receiving topiramate at different doses and placebo. The sample sizes for each group are also mentioned. However, there seems to be some formatting and errors, making parts of the text illegible.*
Label Image (Lbl504360139)

This text describes a medication called topiramate which comes in the form of tablets, with each tablet containing 50mg of the active ingredient. The medication is manufactured by Cadila Healthcare in Ahmedabad, India, and is dispensed in a container. The National Drug Code (NDC) for this medication is 50436-0139-1. The lot number for this medication is 0000 EXPXXRXRX. The medication is exclusively for prescription use and the dosage information is included in the package insert.*
* These product label images have been analyzed using experimental machine learning. Please verify findings with the primary label text.