Product Images Duloxetine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Duloxetine Hydrochloride NDC 50436-2997 by Unit Dose Services, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure1.jpg - Figure1
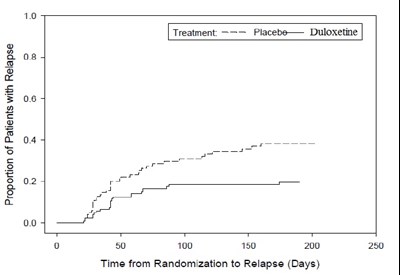
This is a graph showing the proportion of patients with relapse over time from randomization to relapse (in days) for a treatment of placebo versus duloxetine. The x-axis represents the time in days, and the y-axis shows the proportion of patients with relapse. However, without more contextual information, it is unclear what this graph pertains to or what the treatment and relapse refer to.*
Figure2.jpg - Figure2
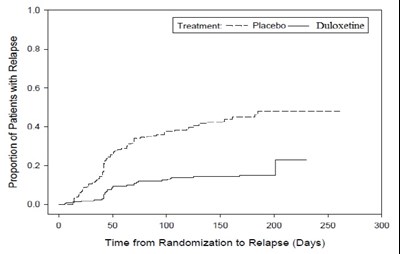
This is a graph showing the proportion of patients with relapse over time after randomization in a clinical trial comparing the treatment groups receiving placebo versus duloxetine. The x-axis represents the time from randomization to relapse in days, and the y-axis shows the proportion of patients with relapse, ranging from 0.06 to 0.1. There are no other details or information available in the given text.*
Figure5.jpg - Figure5
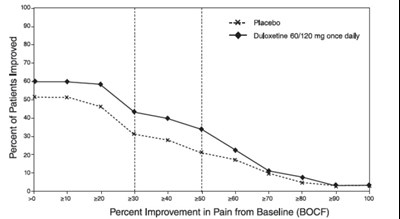
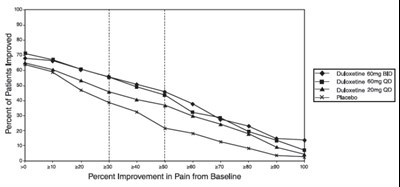
This is a chart or graph showing the percent improvement in pain from the baseline for a treatment that includes placebo and duloxetine doses of 60mg/120mg taken once daily. There are no additional details available.*
Figure7.jpg - Figure7

The text describes the percentage of improvement in patients treated with a placebo and Duloxetine 60/120 mg once daily. The percentage of patients improved is 100. There is also a graph that shows the percent improvement in pain from baseline (BOCF).*
Label Image - lbl504362995

This is a medication called Duloxetine Hydrochloride, with a strength of 20mg and a package size of 60 capsules. The manufacturer is Apotex located in Toronto. The NDC number is 5043629952. It appears to be a delayed-release medication. No further information is available such as usage, indications, or warnings.*
Label Image - lbl504362996

This is a medication for oral use with NDC number 50436-2996-1 containing DULOXETINE HYDROCHLORIDE in delayed-release form. Each capsule has a strength of 30 mg and contains 337 mg of Dioketne Hycrochiorde equal to 30 mg of Duloxtie. It is manufactured by Apotex Inc. in Canada and the lot number is X000 with an expiration date of 30000. The medication is only available through a prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.