Product Images Cialis
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Cialis NDC 50436-4464 by Unit Dose Services, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - cialis uspi f001 v2
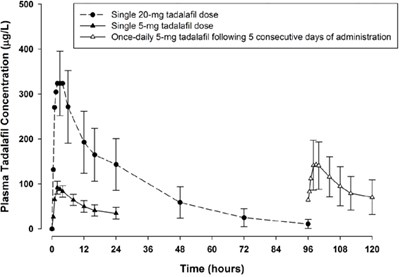
This is a graph showing the plasma concentration of Tadalafil over time, with doses of 20mg and 5mg (taken as a single dose or daily for 5 days) represented by different markers. The concentration of Tadalafil is measured in micrograms per liter (ug/L) and plotted against time in hours. The graph includes a scale from 0 to 500 ug/L and time from 0 to 120 hours.*
Figure - cialis uspi f002 v1
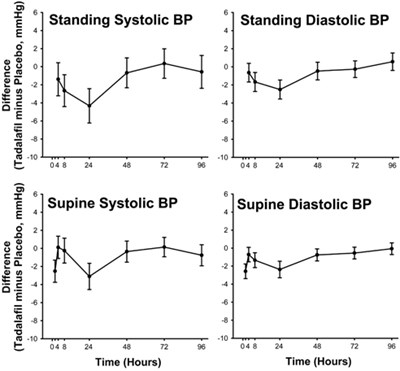
This appears to be a table with headers indicating blood pressure readings for standing and supine positions, with a comparison of Tadalafil and Placebo treatments. The difference in blood pressure (in mmHg) is also indicated. The data is presented over time (in hours).*
Figure 6 - cialis uspi f006 v1
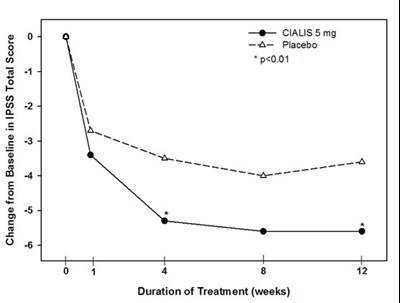
This text provides information on the change from baseline in IPSS total score for patients treated with either CIALIS 5mg or placebo. The graph shows a significant improvement in IPSS total score for those treated with CIALIS 5mg compared to placebo over the duration of treatment in weeks.*
Figure 8 - cialis uspi f008 v1
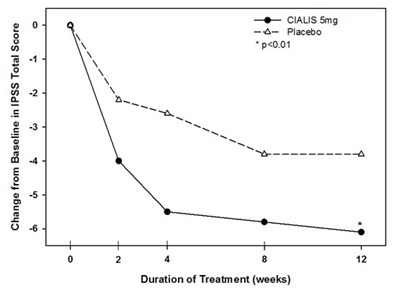
The text describes the change from the baseline in the IPSS (International Prostate Symptom Score) total score after a 12-week treatment period. The treatment included taking 5mg of CIAUS, and a placebo group was also included. The asterisk highlights a statistically significant difference between the CIAUS and placebo groups at a significance level of p<0.01.*
Label Image - lbl504364462

This is a product label for Cialis, which is a medication for treating erectile dysfunction. The NDC code is 50436-4462-1 and the medication comes in 5mg unit-dose tablets. The rest of the text is not clear.*
Label Image - lbl504364463

This is a description of the prescription drug Cialis, specifically the 10 mg tablet form. The NDC number and manufacturer details are provided, along with instructions not to split the tablet and to take the entire dose. The text also includes a warning to keep the drug out of reach and indicates that it is for storage purposes only and requires a prescription.*
Label Image - lbl504364464

NDC: 50436-4464-1 is a medication called CIALIS (Tadalafil) with a dosage of 20 MG / 30 TAB. Each tablet contains 20 mg of tadalafl and should not be split. The entire dose should be taken. Dispense in a tight container and keep out of reach of children while storing at 25°C (77°F). It is a controlled room temperature medication, and the package insert should be referred to for dosage information. The rest of the text seems to be lot numbers and codes for manufacturing purposes and is not useful for a description.*
Label Image - lbl504364465

CIALIS (Tadalafil) is a medication used for once daily use. Each tablet contains 2.5mg of Tadalafil and is manufactured by an unknown company with NDC code 50436-4465-1. The medication should be kept out of reach of children and stored at 25°C. For dosage information, refer to the package insert. There is also information on lot number, expiration date, and serial number provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




