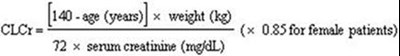Product Images Lyrica
View Photos of Packaging, Labels & Appearance
- Label Image - lbl504366523
- Cockcroft and Gault equation - lyrica 01
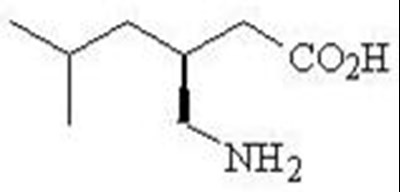
- Chemical Structure - lyrica 02
- Figure 1 - lyrica 03
- Figure 2 - lyrica 04
- Figure 3 - lyrica 05
- Figure 4 - lyrica 06
- Figure 5 - lyrica 07
- Figure 6 - lyrica 08
- Figure 7 - lyrica 09
- Figure 8 - lyrica 10
- Figure 9 - lyrica 11
- Figure 10 - lyrica 12
- Figure 11 - lyrica 13
- Logo - lyrica 14
- lyrica 14a
- lyrica 14b
Product Label Images
The following 17 images provide visual information about the product associated with Lyrica NDC 50436-6523 by Unit Dose Services, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label Image - lbl504366523

This appears to be a label of medication named "Lyrica". It consists of 90 capsules with 75 mg strength. The label also contains some identification codes, including NDC code and WEG NOC. However, some text is not clearly visible or understandable.*
Figure 4 - lyrica 06

The text is a table showing the percent of patients who improved for three different treatments: Pregabalin 200mg three times per day, Pregabalin 100mg three times per day, and a placebo treatment. The table also includes some illegible characters and a reference to "Pl S Rindina."*
Figure 5 - lyrica 07

This text appears to be a table showing the percentage of patients who improved after taking certain medications for pain relief. The table lists two drugs, Pregatalin and Progabalin, with their respective dosages and frequency of use. The third column seems to represent the percentage of improvement in pain from baseline for each medication. However, it is unclear what the measurement scale is for this improvement percentage.*
Figure 8 - lyrica 10

This text seems to be showing different responder rates for some kind of treatment or drug. The rates are displayed in two columns with percentages ranging from 29.4% to 226%. There is also a mention of a placebo and two different dosage levels (2.5 mg/kg/day and 10 mg/kg/day). However, there is not enough context to determine the exact nature of the treatment or drug being evaluated.*
Figure 9 - lyrica 11
This appears to be a medical document discussing the efficacy of Pregabalin at different doses for patient improvement. The only readable information is that 100% of patients improved. The rest of the text is either non-sensical or contains garbled characters.*
Figure 11 - lyrica 13

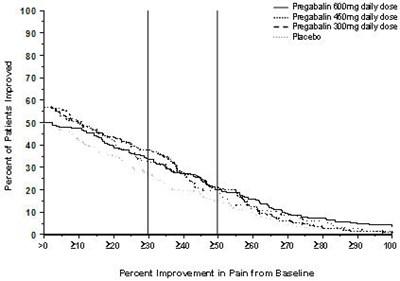
This appears to be a chart or table showing the percentage of subjects who improved after taking Pregabalin, with data points at 50%, 90%, and 100% improvement from a baseline to week 12.*
lyrica 14a

The text provides information about the distribution of products by Parke-Davis, which is a division of Pfizer Inc located in NY, NY 10017.*
lyrica 14b

This is a description of a company that distributes products under the name Parke-Davis, which is a division of Pfizer Inc. The location of Pfizer Inc is in New York, zip code 10017.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.