Product Images Spravato
View Photos of Packaging, Labels & Appearance
Product Label Images
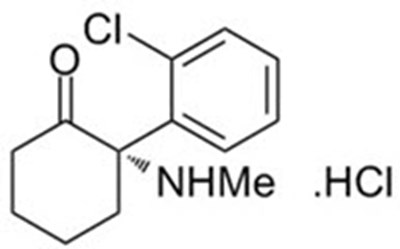
The following 37 images provide visual information about the product associated with Spravato NDC 50458-028 by Janssen Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - spravato 01
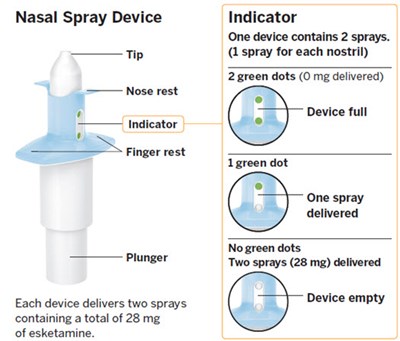
The text describes a nasal spray device with a nose rest, finger rest, plunger, and indicator. Each device contains two sprays of esketamine, totaling 28 mg. The indicator shows the user how many sprays are left in the device with green dots, marking whether the device is full, has one spray left, or is empty.*
Figure - spravato 02

This appears to be a set of instructions for using a medical device. The device seems to come in different dosages, generally measured in milligrams (mg). The instructions suggest that the patient should blow their nose before using the device for the first time only and to confirm the appropriate number of devices needed based on the prescribed dosage.*
Figure - spravato 03

This text provides instructions for a healthcare professional regarding how to prepare a medical device for patient use. They should check the expiration date and not prime the device. If the expiration date is passed, they should get a new device. They should also check that the indicator shows two green dots; if it does not, they should dispose of the device and get a new one. After checking these things, the healthcare professional should hand the device to the patient.*
Figure - spravato 04

This is a set of instructions for administering medication through the nose using a device. The patient is directed to hold the device with their thumb gently supporting the plunger without pressing it. They are further instructed to recline their head at an angle of about 45 degrees during administration to ensure that the medication remains inside the nose.*
Figure - spravato 05
This text describes a medical instruction for a patient, which advises them to spray a medication into each nostril once. The specific medication or purpose is not mentioned.*
Figure - spravato 06

This text appears to be instructions for using a nasal spray medication, including how to properly insert the nozzle into the nostril, breathe in while pushing the plunger, and sniff gently after spraying. It also instructs the patient to switch hands and repeat the process for the second nostril to deliver a second dose.*
Figure - spravato 07

This text provides instructions for a healthcare professional on how to instruct a patient on the proper use of a nasal spray device. The professional is advised to check the indicator on the device and ensure the patient rests for five minutes after each use. It is not clear what medication the device delivers.*
Figure - spravato 08

This text is providing instructions for the disposal of used devices according to the facility procedures and relevant regulations. It specifically mentions the need to adhere to regulations for Schedule II drug products at the federal, state, and local level.*
Figure 1 - spravato 10

This is a population description of individuals of different races aged between 22-50 years, 124-53 years, and with a life expectancy of 75:85. The races include Japanese/White, Chinese/White, and Korean/White. The study provides information on hepatic and renal impairment as well as allergic rhinitis and PK data. However, the text appears to be incomplete and difficult to understand, with some characters not recognized by the system.*
Figure 2 - spravato 11

This text provides a list of drugs and their cytochrome P450 interactions. It also includes information about nasal corticosteroids and decongestant drugs. Additionally, it contains data regarding Cmax and AUC values in reference to intranasal esketamine administration.*
Figure 3 - spravato 12

This is a description of a study on the interactions between certain drugs and liver enzymes. Bupropion is a substrate of hepatic CYP2B6 while midazolam is a substrate of hepatic CYP3A. The study measured Cmax and AUC, and the fold change and 90%C1 were presented graphically. The potential for cytochrome P450 induction by esketamine was also evaluated in the study. Overall, the text appears to be a report of scientific data and findings related to drug interactions and their effects on liver enzymes.*
Figure 4 - spravato 13

The text is not clear and contains a lot of random characters. Hence, the description cannot be generated.*
Figure - spravato 18

This text describes a nasal spray device for esketamine. The device contains 2 sprays and has a plunger. The device has an indicator with green dots to show the number of sprays remaining, with 2 dots indicating a full device and 1 dot indicating one spray has been delivered. Each device delivers a total of 28 mg of esketamine. When the device is empty, it needs to be discarded.*
Figure - spravato 22

This text appears to be a table linking values of weight in milligrams (mg) with the corresponding number of devices. 56 mg corresponds to 2 devices, and 84 mg corresponds to 3 devices.*
Figure - spravato 27

This text describes the method of using a nasal spray - spraying once into each nostril.*
Figure - spravato 35

This text provides important instructions regarding the use and disposal of medication devices. It is emphasized to wait for five minutes after using the device to allow for proper medication absorption. Proper disposal is also highlighted, including following facility procedures for Schedule IIT drugs and adhering to federal, state, and local regulations.*
PRINCIPAL DISPLAY PANEL - Two 28 mg Device Blister Pack Kit - spravato 36

Spravato is a medication for intranasal use only, in the form of a nasal spray containing 56 mg in a dose kit. The medication is meant for patients and contains a medication guide for each patient. The drug should be kept out of reach of children. The product is manufactured by Janssen and is stored at specific temperature conditions. The text also includes some unreadable characters.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.