Product Images Xarelto
View Photos of Packaging, Labels & Appearance
- Figure 1 - xarelto 01
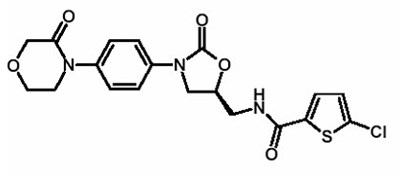
- Chemical Structure - xarelto 02
- Figure 2 - xarelto 03
- Figure 3 - xarelto 04
- Figure 4 - xarelto 05
- Figure 5 - xarelto 06
- Figure 6 - xarelto 07
- Figure 7 - xarelto 08
- Figure 8 - xarelto 09
- PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Label - xarelto 10
- PRINCIPAL DISPLAY PANEL - 15 mg Tablet Bottle Label - xarelto 11
- PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - xarelto 12
- PRINCIPAL DISPLAY PANEL - Kit Carton - xarelto 13
- xarelto 14
- xarelto 15
- xarelto 16
- xarelto 17
- xarelto 18
- xarelto 19
- xarelto 20
- xarelto 21
- xarelto 22
- xarelto 23
- xarelto 24
- xarelto 25
- xarelto 26
- xarelto 27
- xarelto 28
- xarelto 29
- xarelto 30
- xarelto 31
- xarelto 32
- xarelto 33
- xarelto 34
- xarelto 35
- xarelto 36
- xarelto 37
- xarelto 38
- xarelto 39
Product Label Images
The following 39 images provide visual information about the product associated with Xarelto NDC 50458-575 by Janssen Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - xarelto 03
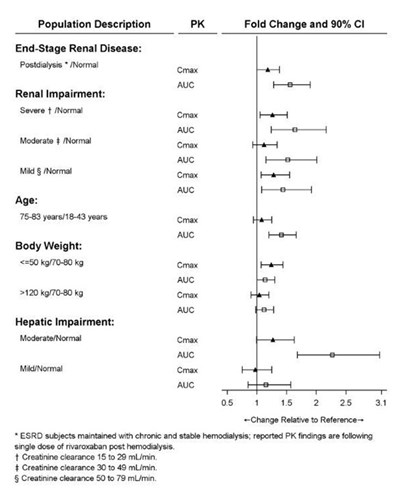
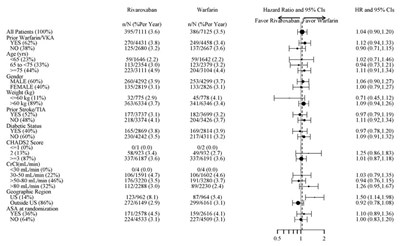
This text contains data on the pharmacokinetics of rivaroxaban in individuals with end-stage renal disease and renal impairment, differentiated by severity, as well as in individuals of different ages and body weights. The data includes peak concentration (Cmax) and area under the curve (AUC) measurements, along with fold change and 90% confidence intervals. The study also delves into the effects of hepatic impairment on the pharmacokinetics of rivaroxaban.*
Figure 3 - xarelto 04

This appears to be a table or list of drugs and their corresponding PK Fold Changes and 90% CI (Confidence Interval) when interacting with other drugs. The interactions are categorized based on whether they are P-gp (P-glycoprotein) or CYP3A (Cytochrome P450 3A) inhibitors or inducers. The table also includes some miscellaneous drugs and their corresponding Cmax (maximum concentration) and Auc (area under the curve) values. The last column shows the change relative to Rivaroxaban alone.*
PRINCIPAL DISPLAY PANEL - 15 mg Tablet Bottle Label - xarelto 11
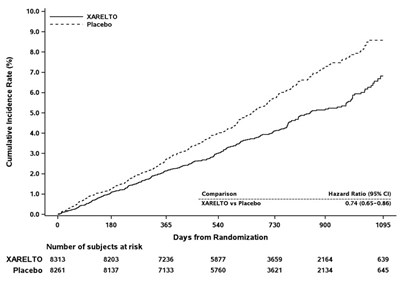
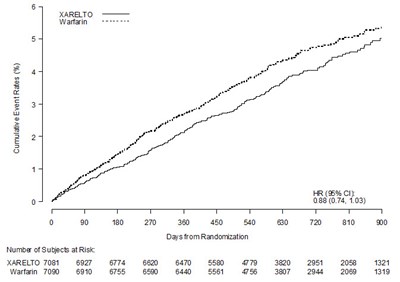
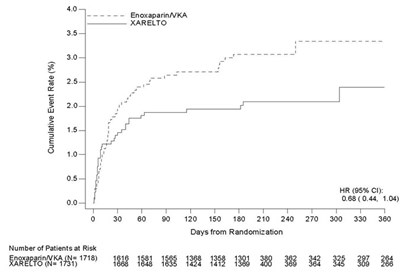
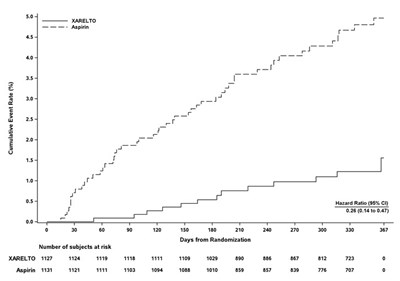
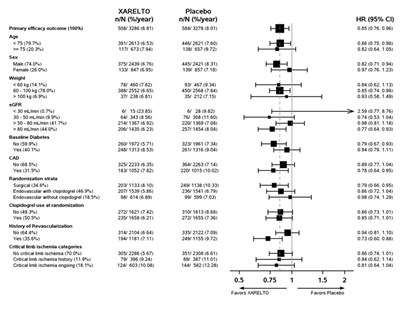
This table represents the results of a study involving a drug called XARELTO compared to placebo. The table shows the cumulative incidence rate (%) at different days from randomization, with the number of subjects at risk for both the drug and the placebo group.*
PRINCIPAL DISPLAY PANEL - 20 mg Tablet Bottle Label - xarelto 12
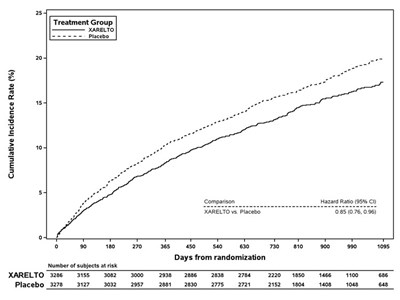
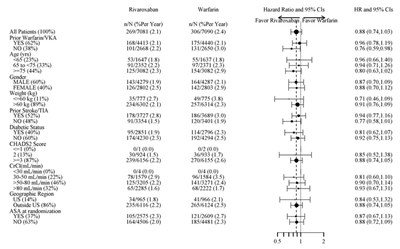
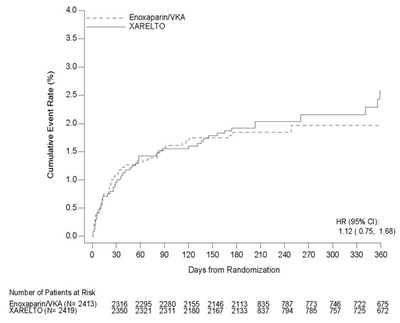
This appears to be a table or chart showing the cumulative incidence rate (%) for two different treatment groups, identified as XARELTO and Placebo. The Hazard Ratio for these groups is also listed as 0.85-0.76, indicating a risk reduction for the XARELTO group. The rest of the text may be indicating time periods, but without context it is difficult to accurately describe.*
xarelto 18

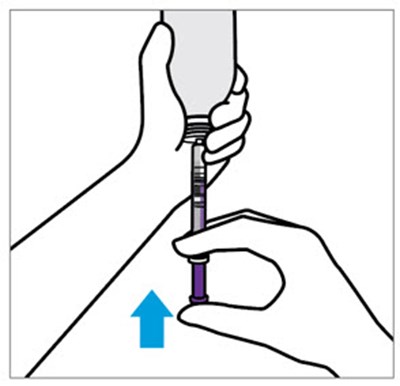
This is a short excerpt from a document that provides instructions for using a bottle adaptor. It advises the reader not to remove the bottle adaptor. Not much additional information can be discerned from this fragment of text.*
xarelto 19
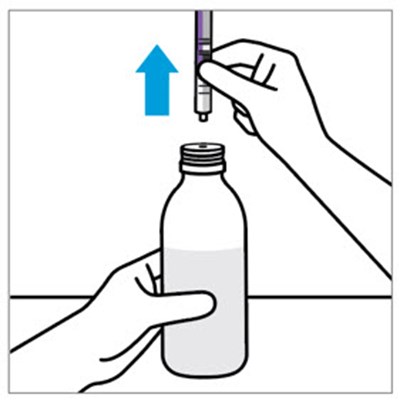
This text appears to be an instruction to check the expiration date of a product. The specific phrase "Discard after" suggests that the product has a limited lifespan and should not be used beyond a certain point for safety or efficacy reasons.*
xarelto 24
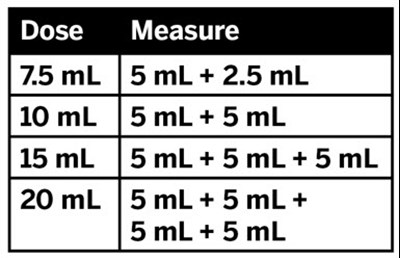
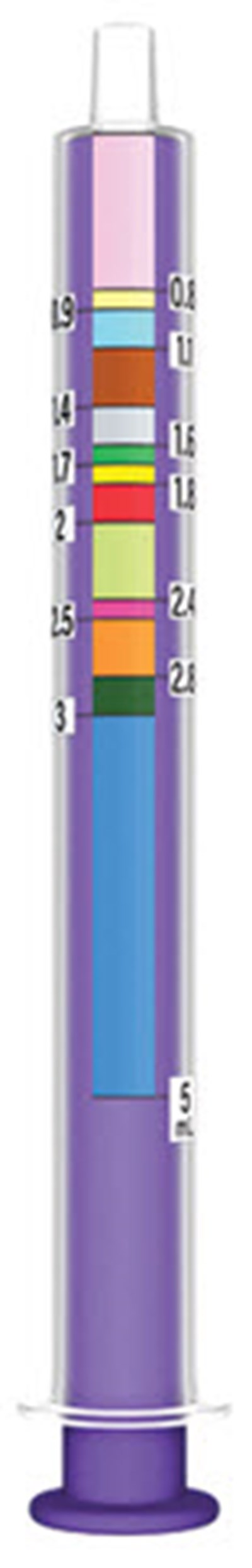
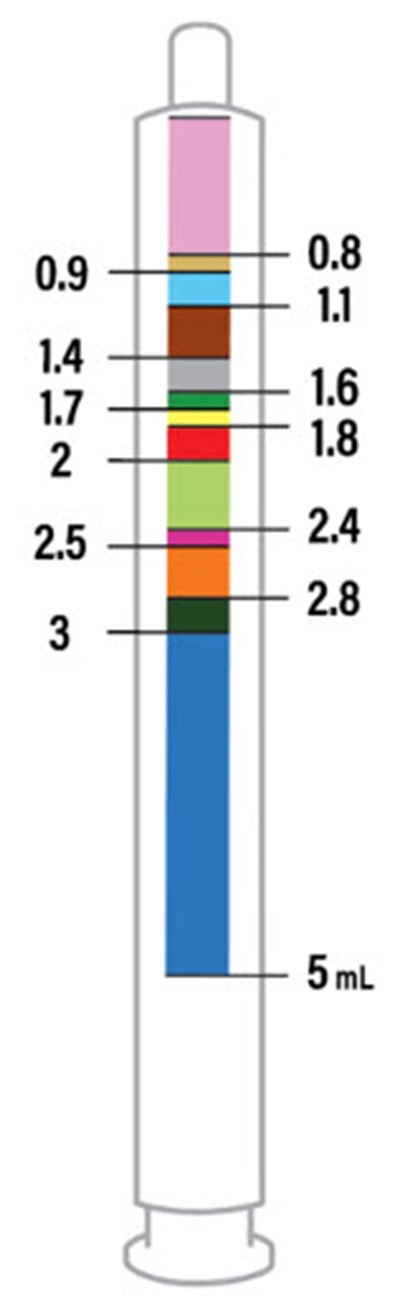
This is a table showing the volume of different solutions. The first column indicates the total volume of the solution, while the second column shows the amounts of two different components added together to make up the total volume. The third column expands on this idea and shows the components of each solution in more detail.*
xarelto 26
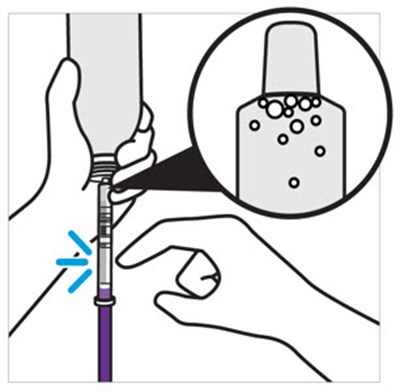
The text contains only two words: "bottle" and "adaptor". It doesn't provide enough context for a specific description.*
xarelto 34
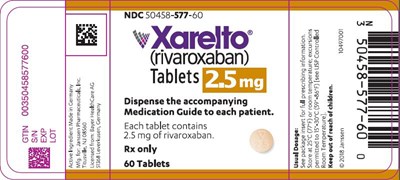
This is the description: This is a medication called Xarelto, with GTIN number 00350458577600 and NDC number 50458-577-60. Each tablet contains 2.5mg of rivaroxaban. It is a prescription medication and 60 tablets are included in the package. A medication guide should be provided to each patient. The text also includes some unreadable characters and is not properly formatted.*
xarelto 36
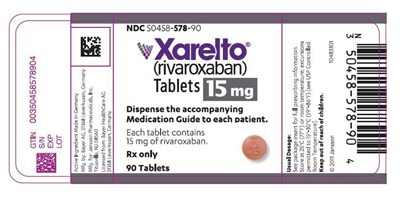
This is a package label for a medication called Xarelto. The medication is in tablet form and contains 15mg of rivaroxaban. The label includes information on dosage, storage instructions, lot number, and expiration date. It also provides the National Drug Code (NDC) and GTIN identification numbers for the medication. Patients should be given a Medication Guide along with the tablets.*
xarelto 37
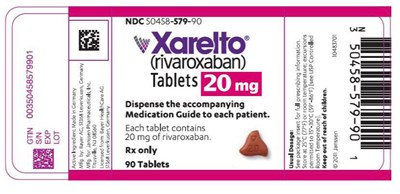
This is a description of the medication Xarelto (rivaroxaban) which comes in a bottle of 90 tablets. Each tablet contains 20 mg of rivaroxaban. A medication guide should be dispensed to each patient along with the tablets. The text also includes an NDC number which could be used for tracking purposes.*
xarelto 38

Xarelto is a medication used for the treatment of deep vein thrombosis and pulmonary embolism. The starter pack provides a 30-day supply, with the first 21 days consisting of a 15mg tablet taken twice a day and the remaining days consisting of a 20mg tablet taken once a day. It is recommended to see a doctor before starting the treatment and to take the medication exactly as prescribed without changing the dose or stopping unless instructed by a doctor. The package contains additional instructions on how to open it and store it properly. This medication should be kept out of reach of children.*
xarelto 39
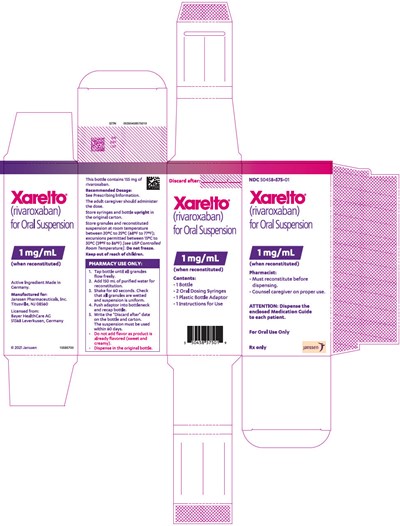
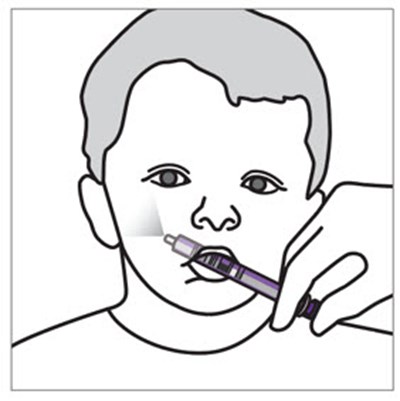
This is a description of Xarelto, a medication with the active ingredient rivaroxaban for oral suspension. The bottle contains 155mg of rivaroxaban, which is made in Germany and manufactured for Janssen Pharmaceuticals. The dosage and administration instructions are provided in the prescribing information, and the suspension must be used within 60 days once reconstituted. The pharmacist must reconstitute the medication before dispensing and counsel the caregiver on its proper use. The medication should be stored at room temperature and kept out of reach of children. The rest of the text explains how to reconstitute the suspension using a plastic bottle adapter and dosing syringes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.