FDA Label for Good Care Surgical Brush With Nail Cleaner
View Indications, Usage & Precautions
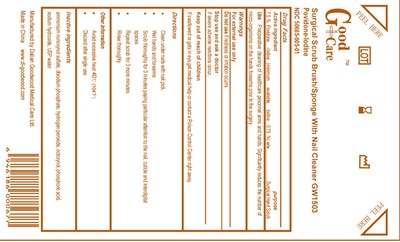
Good Care Surgical Brush With Nail Cleaner Product Label
The following document was submitted to the FDA by the labeler of this product Dalian Goodwood Medical Care Ltd.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Active Ingredient
Povidone Iodine USP, 7.5% w/v
(minimum available Iodine 0.75%) w/w
Otc - Purpose
Purpose
Surgical Hand Scrub
Indications & Usage
Use
Preoperative cleaning of healthcare personnel arms and hands. Significantly reduces the number of micro-organisms on the hands forearms prior to the surgery.
Warnings
Warnings
For external use only.
Otc - Do Not Use
Do not use if redness or irritation ocurrs
Otc - Stop Use
Stop use and ask a doctor if severe adverse reactions occur.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed or gets in eyes, get medical help or contact a Poison Control Center right away.
Dosage & Administration
Directions
- Clean under nails with nail pick
- Wet hands and forearms
- Scrub thoroughly for 3 minutes paying particular attention to the nail, cuticle and interdigital spaces
- Repeat scrub for 3 more minutes
- Rinse thoroughly
Storage And Handling
Other information
- Avoid excessive heat 40 degrees C (104 degrees F)
- Discard after single use
Inactive Ingredient
Inactive ingredients
ammonium nonoxynol sulfate, disodium phosphate, hydrogen peroxide, octoxynol, phosphoric acid, sodium hydroxide, USP water
Other
Manufactured by DALIAN GOODWOOD MEDICAL CARE LTD.
Made in China www.dl-goodwood.com
Package Label.Principal Display Panel
Good CareSurgical Scrub Brush/ Sponge With Nail Cleaner GW1503Povidone-Iodine
* Please review the disclaimer below.