Product Images Flunisolide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Flunisolide NDC 50742-317 by Ingenus Pharmaceuticals, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
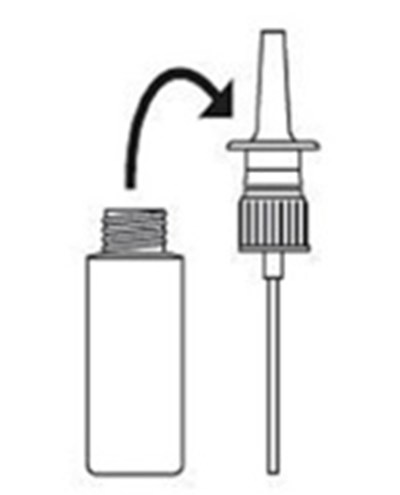
figure - figure001

This text describes the parts of a nasal spray unit, including the preassembled pump, a tip, finger rests, and directions to spray and cover the area.*
Label - image0002

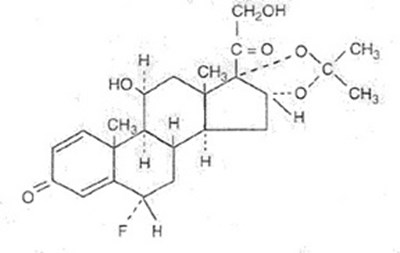
Flunisolide Nasal Solution is a prescription medication intended for intranasal use only. Each spray delivers 25mcg flunisolide. The solution contains active ingredients such as Flunisolide 0.25mg (0.025%) and other inactive ingredients such as Propylene Glycol, Polyethylene Glycol 3350, Butylated Hydroxyanisole, Edetate Disodium, Sodium Citrate, Citric Acid, and Purified Water. Sodium Hydroxide and/or Hydrochloric Acid may be added to adjust pH (5.1 - 5.4). The medicine is available in 25mL with 200 metered sprays. Refer to the package insert for complete prescribing information and instructions for using the nasal pump. The solution should be stored at 20° to 25°C (68° to 77°F), with excursions permitted between 15° to 30°C (59° to 86°F). Keep it out of reach of children.*
Carton - image001

This appears to be a product label for Flunisolide Nasal Solution, indicating its ingredients, dosage, and usage instructions. It also mentions important warnings to keep the product out of reach of children and instructions to read before use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.