Product Images Irinotecan Hydrochloride
View Photos of Packaging, Labels & Appearance
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 01
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 02
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 03
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 04
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 05
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 06
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 07
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 08
- 27b29c94 6029 4c8d 9cc9 845b2d5cb006 09
Product Label Images
The following 9 images provide visual information about the product associated with Irinotecan Hydrochloride NDC 50742-401 by Ingenus Pharmaceuticals, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
27b29c94 6029 4c8d 9cc9 845b2d5cb006 03
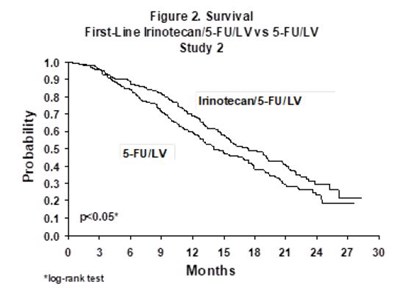
This is a graph for a study (Study 2) comparing the survival rate between two treatments: Irinotecan/5-FU/LV and 5-FULV. The x-axis shows the number of months after treatment, and the y-axis represents the probability of survival. The log-rank test was used to compare the two treatments.*
27b29c94 6029 4c8d 9cc9 845b2d5cb006 04
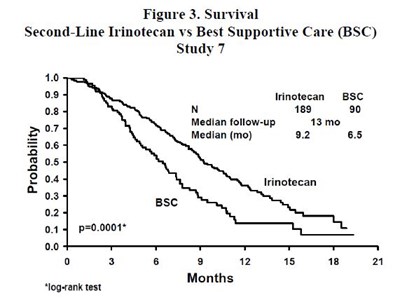
This is a figure showing survival data for a study (Study 7) comparing second-line Irinotecan treatment to Best Supportive Care (BSC) for patients with a certain condition. The figure displays the number of patients in each group (189 for Irinotecan and 90 for BSC), the median follow-up time (13 months), and the median survival time (9.2 months for Irinotecan and 6.5 months for BSC). Additionally, the figure shows a statistical test (log-rank) with a p-value of 0.0001 indicating a significant difference in survival between the two treatments.*
27b29c94 6029 4c8d 9cc9 845b2d5cb006 09

This is a product description of Irinotecan Hydrochloride Injection, USP, a cytotoxic agent used for intravenous use. The 100mg/5mL single-dose vial must be diluted before use. The product is manufactured by Ingenus Pharmaceuticals and comes in a 2mg/mL concentration, and should be stored properly to prevent freezing. Contact information for the manufacturer is provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





